5-vinyluracil
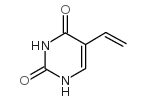
5-vinyluracil structure
|
Common Name | 5-vinyluracil | ||
|---|---|---|---|---|
| CAS Number | 37107-81-6 | Molecular Weight | 138.12400 | |
| Density | 1.332g/cm3 | Boiling Point | N/A | |
| Molecular Formula | C6H6N2O2 | Melting Point | >300ºC (dec.)(lit.) | |
| MSDS | Chinese USA | Flash Point | N/A | |
| Symbol |

GHS07 |
Signal Word | Warning | |
|
A method for the rapid preparation of 5-vinyluracil in high yield.
Nucleic Acids Res. 1(1) , 105-7, (1974) A method for the rapid preparation of the thymineanalogue, 5-vinyluracil, in 83% yield from 5-(1-hydroxyethyl)uracil via the methanesulphonyl ester is reported. |
|
|
Design and synthesis of the tumor-activated prodrug of dihydropyrimidine dehydrogenase (DPD) inhibitor, RO0094889 for combination therapy with capecitabine.
Bioorg. Med. Chem. Lett. 13(5) , 867-72, (2003) A series of tumor-activated prodrugs of the inhibitors of dihydropyrimidine dehydrogenase (DPD), an enzyme catabolizing 5-fluorouracil (5-FU: 4g), has been designed and synthesized. RO0094889 (11c) is a prodrug of 5-vinyluracil (4c), a known DPD inhibitor, an... |
|
|
Incorporation of 5-substituted uracil derivatives into nucleic acids. The isolation and gamma-radiation-sensitivity of bacteriophage T3 containing the thymine analogue 5-vinyluracil.
Biochem. J. 187(1) , 257-60, (1980) Bacteriophage T3 was produced in a form that contained 32% of its normal DNA thymine residues replaced with 5-vinyluracil residues by infecting a thymine-requiring strain of Escherichia coli with phage T3 in a medium containing 5-vinyluracil. When 2'-deoxy-5-... |