D-3-Deoxyglucosone
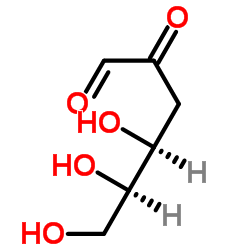
D-3-Deoxyglucosone structure
|
Common Name | D-3-Deoxyglucosone | ||
|---|---|---|---|---|
| CAS Number | 4084-27-9 | Molecular Weight | 162.141 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 400.1±45.0 °C at 760 mmHg | |
| Molecular Formula | C6H10O5 | Melting Point | 73-75ºC | |
| MSDS | Chinese USA | Flash Point | 209.9±25.2 °C | |
|
3-Deoxyglucosone: a potential glycating agent accountable for structural alteration in H3 histone protein through generation of different AGEs.
PLoS ONE 10(2) , e0116804, (2015) Advanced glycation end-products (AGEs) are heterogeneous group of compounds, known to be implicated in diabetic complications. One of the consequences of the Maillard reaction is attributed to the production of reactive intermediate products such as α-oxoalde... |
|
|
Studies on the Formation of Maillard and Caramelization Products from Glucosamine Incubated at 37 °C.
J. Agric. Food Chem. 63 , 6249-61, (2015) This experiment compared the in vitro degradation of glucosamine (GlcN), N-acetylglucosamine, and glucose in the presence of NH3 incubated at 37 °C in phosphate buffer from 0.5 to 12 days. The reactions were monitored with UV-vis absorption and fluorescence e... |
|
|
GC-MS Method for the Quantitation of Carbohydrate Intermediates in Glycation Systems.
J. Agric. Food Chem. 63 , 5911-9, (2015) Glycation is a ubiquitous nonenzymatic reaction of carbonyl compounds with amino groups of peptides and proteins, resulting in the formation of advanced glycation end-products (AGEs) and thereby affecting the properties and quality of thermally processed food... |
|
|
Upregulation of galectin-3 in immortalized Schwann cells IFRS1 under diabetic conditions.
Neurosci. Res. 92 , 80-5, (2015) A spontaneously immortalized adult Fischer rat Schwann cell line IFRS1 retains the characteristic features of normal Schwann cells, and can be a useful tool for the study of diabetic neuropathy. In the present study, we examined the effects of high glucose an... |
|
|
Reduction of lipid peroxidation products and advanced glycation end-product precursors by cyanobacterial aldo-keto reductase AKR3G1—a founding member of the AKR3G subfamily.
FASEB J. 29(1) , 263-73, (2015) The purpose of this study was to investigate the origin and function of the aldo-keto reductase (AKR) superfamily as enzymes involved in the detoxification of xenobiotics. We used the cyanobacterium Synechocystis sp. PCC 6803 as a model organism and sequence ... |
|
|
Formation of alpha-dicarbonyl compounds in beer during storage of Pilsner.
J. Agric. Food Chem. 56(11) , 4134-44, (2008) With the aim of determining the formation of alpha-dicarbonyl intermediates during beer aging on the shelf, alpha-dicarbonyls were identified and quantified after derivatization with 1,2-diaminobenze to generate quinoxalines. The sensory effects of alpha-dica... |
|
|
Rapid and sensitive determination of the intermediates of advanced glycation end products in the human nail by ultra-performance liquid chromatography with electrospray ionization time-of-flight mass spectrometry.
Anal. Biochem. 424(2) , 187-94, (2012) The resolution of the intermediate advanced glycation end products (AGEs) in the human nail was carried out by the combination of 4,5-dimethyl-1,2-phenylenediamine (DMPD) derivatives and ultra-performance liquid chromatography with electrospray ionization tim... |
|
|
Survey of 1,2-dicarbonyl compounds in commercial honey of different floral origin.
J. Food Sci. 76(8) , C1203-10, (2011) In this study we conducted a survey of the concentrations of the major 1,2-dicarbonyl compounds in 40 commercial honey samples from 12 different floral origins. 3-Deoxyglucosone (3-DG), glyoxal (GO), and methylglyoxal (MGO) were measured, using their correspo... |
|
|
Quantification of the six major α-dicarbonyl contaminants in peritoneal dialysis fluids by UHPLC/DAD/MSMS.
Anal. Bioanal. Chem 401 , 1183-1193, (2011) During heat sterilization of peritoneal dialysis solutions, glucose is partially transformed into glucose degradation products (GDPs), which significantly reduce the biocompatibility of these medicinal products. Targeted α-dicarbonyl screening identified glyo... |
|
|
Identification and determination of alpha-dicarbonyl compounds formed in the degradation of sugars.
Biosci. Biotechnol. Biochem. 71(10) , 2465-72, (2007) The alpha-dicarbonyl compounds formed in the degradation of glucose and fructose were analyzed by HPLC using 2,3-diaminonaphthalene as derivatizing reagent, and identified as glucosone (GLUCO), 3-deoxyglucosone (3DG), 3-deoxyxylosone (3DX), tetrosone (TSO), t... |