o-(2-Benzothiazolyl)phenol
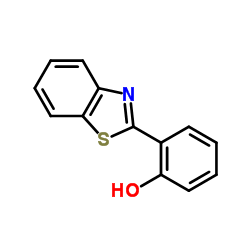
o-(2-Benzothiazolyl)phenol structure
|
Common Name | o-(2-Benzothiazolyl)phenol | ||
|---|---|---|---|---|
| CAS Number | 3411-95-8 | Molecular Weight | 227.282 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 402.2±47.0 °C at 760 mmHg | |
| Molecular Formula | C13H9NOS | Melting Point | 128-132 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 197.0±29.3 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
|
Modulation of firefly luciferase stability and impact on studies of gene regulation.
Gene 103(2) , 171-7, (1991) Two of the reporter enzymes most commonly used in studies of eukaryotic gene expression are chloramphenicol acetyl-transferase (CAT) and firefly luciferase (Luc). CAT has a half-life of about 50 h in mammalian cells, making it useful for transient transfectio... |
|
|
The effect of hydrogen bonding on the excited-state proton transfer in 2-(2'-hydroxyphenyl)benzothiazole: a TDDFT molecular dynamics study.
Phys. Chem. Chem. Phys. 14(25) , 9016-25, (2012) The dynamics of the excited-state proton transfer (ESPT) in a cluster of 2-(2'-hydroxyphenyl)benzothiazole (HBT) and hydrogen-bonded water molecules was investigated by means of quantum chemical simulations. Two different enol ground-state structures of HBT i... |
|
|
A highly selective fluorescent probe for fast detection of hydrogen sulfide in aqueous solution and living cells.
Chem. Commun. (Camb.) 48(88) , 10871-3, (2012) A new ratiometric fluorescence probe E1 based on an excited-state intramolecular proton transfer (ESIPT) mechanism for detection of hydrogen sulfide (H(2)S) is reported. E1 responds to H(2)S quickly and showed a 30-fold fluorescence enhancement in 2 minutes. ... |
|
|
Accessing the long-lived triplet excited states in bodipy-conjugated 2-(2-hydroxyphenyl) benzothiazole/benzoxazoles and applications as organic triplet photosensitizers for photooxidations.
J. Org. Chem. 77(14) , 6166-78, (2012) Bodipy derivatives containing excited state intramolecular proton transfer (ESIPT) chromophores 2-(2-hydroxyphenyl) benzothiazole and benzoxazole (HBT and HBO) subunits were prepared (7-10). The compounds show red-shifted UV-vis absorption (530-580 nm; ε up t... |
|
|
New excited state intramolecular proton transfer (ESIPT) dyes based on naphthalimide and observation of long-lived triplet excited states.
Chem. Commun. (Camb.) 48(78) , 9720-2, (2012) A new excited state intramolecular proton transfer chromophore of naphthalimide (NI) conjugated 2-(2-hydroxyphenyl) benzothiazole (HBT) was prepared which shows red shifted absorption and long-lived triplet excited states. |
|
|
Active role of proton in excited state intramolecular proton transfer reaction.
J. Phys. Chem. A 117(7) , 1400-5, (2013) Proton transfer is one of the most important elementary reactions in chemistry and biology. The role of proton in the course of proton transfer, whether it is active or passive, has been the subject of intense investigations. Here we demonstrate the active ro... |
|
|
Theoretical studies of molecular structure, electronic structure, spectroscopic properties and the ancillary ligand effect: a comparison of tris-chelate ML3-type and ML2X-type species for gallium(III) complexes with N,O-donor phenolic ligand, 2-(2-hydroxyphenyl)benzothiazole.
Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 78(2) , 809-17, (2011) Two Ga(III) complexes with main ligand, 2-(2-hydroxyphenyl)benzothiazole (HL'), namely mixed-ligand ML2X-type [GaL'2X'] (1) (HX'=acetic acid, as ancillary ligand) and the meridianal tris-chelate [GaL'3] (2) have been investigated by the density functional the... |