felbamate
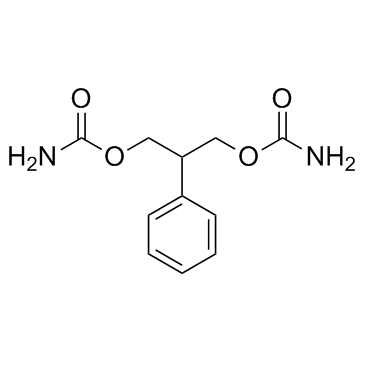
felbamate structure
|
Common Name | felbamate | ||
|---|---|---|---|---|
| CAS Number | 25451-15-4 | Molecular Weight | 238.240 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 511.9±50.0 °C at 760 mmHg | |
| Molecular Formula | C11H14N2O4 | Melting Point | 148-1500C | |
| MSDS | Chinese USA | Flash Point | 288.4±26.4 °C | |
| Symbol |



GHS02, GHS06, GHS08 |
Signal Word | Danger | |
|
Pediatric super-refractory status epilepticus treated with allopregnanolone.
Ann. Neurol. 76(6) , 911-5, (2014) Super-refractory status epilepticus is a life-threatening condition. Resistance to benzodiazepine and barbiturate treatment for this disorder is thought to be due to internalization of synaptic γ-aminobutyric acid (GABA)A receptors, and withdrawal of benzodia... |
|
|
Enhanced glutamatergic transmission reduces the anticonvulsant potential of lamotrigine but not of felbamate against tonic-clonic seizures.
Pharmacol. Rep. 59(4) , 462-6, (2007) The efficacy of lamotrigine and felbamate against maximal electroshock (MES)-induced seizures was assessed under conditions mimicking the pharmacoresistance associated with an increased excitatory neurotransmission. N-methyl-D-aspartate (NMDA), but not kainat... |
|
|
Behavioral and cognitive effects of anti-epileptic drugs.
Discov. Med. 9(45) , 138-44, (2010) Anti-epileptic drugs (AEDs) have a variety of mechanisms of action which are reflected through different anticonvulsant activities and behavioral effects. Two categories of AEDs are considered based on psychotropic profile. The first group is characterized by... |
|
|
Felbamate in epilepsy therapy: evaluating the risks.
Drug Saf. 21(3) , 225-39, (1999) Felbamate demonstrates a unique therapeutic profile and often results in seizure control when other agents fail. Its use has been associated with risks for aplastic anaemia and hepatic failure. A number of confounding factors makes the actual incidence rate f... |
|
|
Simultaneous HPLC-UV analysis of rufinamide, zonisamide, lamotrigine, oxcarbazepine monohydroxy derivative and felbamate in deproteinized plasma of patients with epilepsy.
J. Chromatogr. B. Analyt. Technol. Biomed. Life Sci. 878(3-4) , 461-5, (2010) We present an implementation of a method we previously reported allowing the newer antiepileptic drugs (AEDs) rufinamide (RFN) and zonisamide (ZNS) to be simultaneously determined with lamotrigine (LTG), oxcarbazepine's (OXC) main active metabolite monohydrox... |
|
|
Felbamate.
Epilepsia 40 Suppl 5 , S57-62, (1999) Felbamate (FBM) was the first of the new antiepileptic drugs (AEDs) approved in the United States in 1993 with broad-spectrum efficacy against partial and generalized seizures of various types, and indicated for use as adjunctive and monotherapy. The identifi... |
|
|
Characterization of the gating conformational changes in the felbamate binding site in NMDA channels.
Biophys. J. 93(2) , 456-66, (2007) The anticonvulsant effect of felbamate (FBM) is ascribable to inhibition of N-methyl-d-aspartate (NMDA) currents. Using electrophysiological studies in rat hippocampal neurons to examine the kinetics of FBM binding to and unbinding from the NMDA channel, we s... |
|
|
Felbamate.
Epilepsia 36 Suppl 2 , S66-72, (1995) After the first year of clinical experience, felbamate (FBM) appears to be a valuable antiepileptic drug (AED) for the treatment of intractable epilepsy. However, many patients experience side effects that may discourage continued usage. These may be decrease... |
|
|
NMDA receptor antagonist felbamate reduces behavioral deficits and blood-brain barrier permeability changes after experimental subarachnoid hemorrhage in the rat.
J. Neurotrauma 24(4) , 732-44, (2007) Increased levels of glutamate and aspartate have been detected after subarachnoid hemorrhage (SAH) that correlate with neurological status. The NMDA receptor antagonist felbamate (FBM; 2-phenyl-1,3-propanediol dicarbamate) is an anti-epileptic drug that elici... |
|
|
Mechanisms of idiosyncratic drug reactions: the case of felbamate.
Chem. Biol. Interact. 142(1-2) , 99-117, (2002) Idiosyncratic drug reactions (IDR) are a specific type of drug toxicity characterized by their delayed onset, low incidence and reactive metabolite formation with little, if any, correlation between pharmacokinetics or pharmacodynamics and the toxicological o... |