t-Butylbenzene
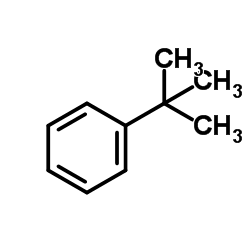
t-Butylbenzene structure
|
Common Name | t-Butylbenzene | ||
|---|---|---|---|---|
| CAS Number | 98-06-6 | Molecular Weight | 134.218 | |
| Density | 0.9±0.1 g/cm3 | Boiling Point | 167.8±7.0 °C at 760 mmHg | |
| Molecular Formula | C10H14 | Melting Point | -58 °C | |
| MSDS | Chinese USA | Flash Point | 34.4±0.0 °C | |
| Symbol |


GHS02, GHS07 |
Signal Word | Warning | |
|
QSPR modeling of octanol/water partition coefficient for vitamins by optimal descriptors calculated with SMILES.
Eur. J. Med. Chem. 43 , 714-40, (2008) Simplified molecular input line entry system (SMILES) has been utilized in constructing quantitative structure-property relationships (QSPR) for octanol/water partition coefficient of vitamins and organic compounds of different classes by optimal descriptors.... |
|
|
Evaluation of injection methods for fast, high peak capacity separations with low thermal mass gas chromatography.
J. Chromatogr. A. 1392 , 82-90, (2015) Low thermal mass gas chromatography (LTM-GC) was evaluated for rapid, high peak capacity separations with three injection methods: liquid, headspace solid phase micro-extraction (HS-SPME), and direct vapor. An Agilent LTM equipped with a short microbore capil... |
|
|
Development of a living membrane comprising a functional human renal proximal tubule cell monolayer on polyethersulfone polymeric membrane.
Acta Biomater. 14 , 22-32, (2015) The need for improved renal replacement therapies has stimulated innovative research for the development of a cell-based renal assist device. A key requirement for such a device is the formation of a "living membrane", consisting of a tight kidney cell monola... |
|
|
Metabolites of three structural isomers of butylbenzene in the bile of rainbow trout.
Bull. Environ. Contam. Toxicol. 44(3) , 487-93, (1990)
|
|
|
Large Raman-scattering activities for the low-frequency modes of substituted benzenes: induced polarizability and stereo-specific ring-substituent interactions.
J. Chem. Phys. 124(10) , 104301, (2006) The large nonresonant Raman-scattering activities of the out-of-plane bending and torsional modes of monosubstituted benzene analogs are studied by low-frequency Raman experiments and B3LYP6-31++G(d,p) calculations. Electronic interactions between the sigma o... |
|
|
Covalent bulk functionalization of graphene.
Nature Chemistry 3(4) , 279-86, (2011) Graphene, a truly two-dimensional and fully π-conjugated honeycomb carbon network, is currently evolving into the most promising successor to silicon in micro- and nanoelectronic applications. However, its wider application is impeded by the difficulties in o... |
|
|
Effect of solvent on the lithium-bromine exchange of aryl bromides: reactions of n-butyllithium and tert-butyllithium with 1-bromo-4-tert-butylbenzene at 0 degrees C.
J. Org. Chem. 71(7) , 2825-8, (2006) The outcome of reactions of 1-bromo-4-tert-butylbenzene (1), a representative aryl bromide, with n-BuLi or t-BuLi at 0 degrees C in a variety of solvent systems has been investigated. The products of reactions of 1 with n-BuLi vary significantly with changes ... |
|
|
Biodegradation of alkyl derivatives of aromatic hydrocarbons and cell surface properties of a strain of Pseudomonas stutzeri.
Chemosphere 90(2) , 471-8, (2013) Pseudomonas stutzeri strain 9 was isolated from petroleum-contaminated soil. The main purpose of this study was to investigate how the long-term contact of this strain with diesel oil influences its surface and biodegradation properties. The experiments showe... |
|
|
A computational study of tert-butylbenzenium ions.
J. Phys. Chem. A 115(14) , 3106-15, (2011) A computational study of tert-butylbenzenium ions has been performed. Structures and energies of the various isomers and the transition states for their interconversions have been determined. The existence of a stable π-electron complex (called A1) between a ... |
|
|
Cellulose tris(3,5-dimethylphenylcarbamate)-coated zirconia as a chiral stationary phase for HPLC.
Anal. Chem. 71(15) , 3013-21, (1999) In this work we explore the use of microparticulate porous zirconia coated with cellulose tris(3,5-dimethylphenyl-carbamate) (CDMPC) as a support for separation of chiral compounds by HPLC. The surface of zirconia, previously sintered but not rehydroxylated, ... |