Vanillyl methyl ketone
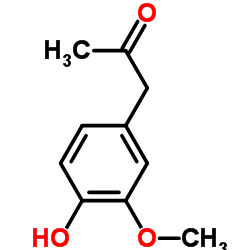
Vanillyl methyl ketone structure
|
Common Name | Vanillyl methyl ketone | ||
|---|---|---|---|---|
| CAS Number | 2503-46-0 | Molecular Weight | 180.201 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 305.6±27.0 °C at 760 mmHg | |
| Molecular Formula | C10H12O3 | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 119.7±17.2 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
|
Analysis of 3,4-methylenedioxymethamphetamine (MDMA) and its metabolites in plasma and urine by HPLC-DAD and GC-MS.
J. Anal. Toxicol. 20(6) , 432-40, (1996) In Europe, the compound 3,4-methylenedioxymethamphetamine (MDMA, Ecstasy, Adam), in addition to cannabis, is the most abused illicit drug at all-night "techno" parties. Methods for the determination of MDMA and its metabolites, 4-hydroxy-3-methoxymethamphetam... |
|
|
Analysis of lignocellulose derived phenolic monomers by headspace solid-phase microextraction and gas chromatography.
J. Chromatogr. A. 1307 , 144-57, (2013) A headspace solid-phase microextraction method with subsequent GC-MS (HS-SPME/GC-MS) was established for the quantitative analysis of volatile lignin derived phenolic monomers in complex aqueous solutions. Extraction was done using a polyacrylate fiber. The o... |
|
|
The effect of 3,4-methylenedioxymethamphetamine (MDMA, 'ecstasy') and its metabolites on neurohypophysial hormone release from the isolated rat hypothalamus.
Br. J. Pharmacol. 135(3) , 649-56, (2002) Methylenedioxymethamphetamine (MDMA, 'ecstasy'), widely used as a recreational drug, can produce hyponatraemia. The possibility that this could result from stimulation of vasopressin by MDMA or one of its metabolites has been investigated in vitro. Release of... |
|
|
Metabolism of 1-(4-hydroxy-3-methoxyphenyl)-2-propanone, a smoke flavour ketone, in rat.
Xenobiotica 23(6) , 693-701, (1993) 1. Metabolites of 1-(4-hydroxy-3-methoxyphenyl)-2-propanone (HMP-one), a smoke flavour compound, were isolated from rat urine using hydrolysis, ether extraction, t.l.c. and g.l.c. 2. Three metabolites were identified by mass spectrometry and independent synth... |
|
|
Aqueous-phase hydrodeoxygenation of bio-derived phenols to cycloalkanes. Zhao C, et al.
J. Catal. 280(1) , 8-16, (2011)
|