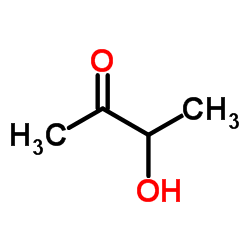
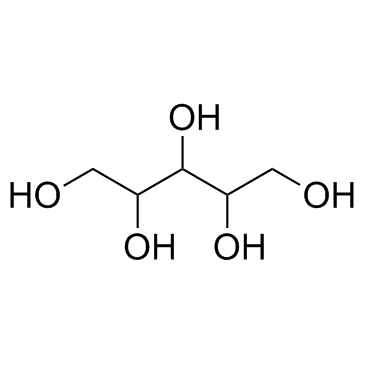
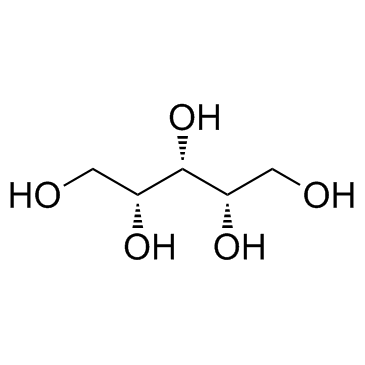
488-81-3
| Name | D-ribitol |
|---|---|
| Synonyms |
(2R,3s,4S)-1,2,3,4,5-Pentanpentol
adonitol ADONIT ADONITE ADONITOL(RG) (2R,3s,4S)-pentane-1,2,3,4,5-pentol RIBITO Adonito D-erythro-Pentitol MFCD00064291 D-Ribitol meso ribitol Ribitol (2R,3s,4S)-1,2,3,4,5-Pentanepentol Adonite Ribitol EINECS 207-685-7 Adonite,Ribitol meso-ribitol D-(+)-Arabitol Adonitrol |
| Description | Ribitol is a crystalline pentose alcohol formed by the reduction of ribose. Enhancing the flux of D-glucose to the pentose phosphate pathway in Saccharomyces cerevisiae for the production of D-ribose and ribitol. |
|---|---|
| Related Catalog | |
| Target |
Human Endogenous Metabolite |
| In Vitro | Ribitol is a reduced sugar[1]. Phosphoglucose isomerase-deficient (pgi1) strains of Saccharomyces cerevisiae are studied for the production of D-ribose and Ribitol from D-glucose via the intermediates of the pentose phosphate pathway. Overexpression of the gene encoding sugar phosphate phosphatase (DOG1) of S. cerevisiae is needed for the production of D-ribose and Ribitol. The engineered strains are compared for their ability to produce the PPP-derived 5-carbon compounds Ribitol and D-ribose from D-glucose[2]. |
| Kinase Assay | The high-performance liquid chromatography (HPLC) analyses are carried out using a Fast Acid Column (100×7.8 mm) and a HPX-87H Ion Exclusion Column (300 mm×7.8 mm) in series with 2.5 mM H2SO4 in water as the mobile phase at a flow rate of 0.3 mL/min, at 55°C. This method enabled quantification of D-glucose, ethanol, glycerol, D-xylulose, Ribitol, and xylitol. D-ribose, D-ribulose, and D-arabitol coeluted on the Aminex HPX-87H column. The CarboPac MA-1 column of Dionex ICS-3000 is used to analyze representative culture supernatant samples for the presence of arabitol and xylitol. Samples are run at column temperature of 30°C with 480 mM NaOH at flow rate 0.4 mL/min. The CarboPac MA-1 column separated D-arabitol from D-ribose and D-ribulose, but the alkaline conditions degraded D-ribulose interfering with the quantification of D-ribose.Yeast cells are disrupted with glass beads in 100 mM sodium phosphate buffer pH 7.0 containing phenylmethylsulfonyl fluoride and pepstatin A in final concentrations of 0.17 mg/mL and 0.01 mg/mL, respectively.The activity of NAD+-dependent Gdh2p is measured in a reaction buffer of 0.5 M triethanol amine pH 7.7 and 2 mM NADH. After addition of the cell lysate, the reaction is started by adding a mixture of α-ketoglutarate (100 mM) and NH4Cl (200 mM) to a final concentration of 2.4 mM and 4.9 mM, respectively. The GapB activity is measured. Shortly, the reaction mixture is 500 mM triethanol amine pH 7.8, 1 mM ATP, 2 mM MgCl2, 200 μM NADPH, and 10 μg/mL of phosphoglycerate kinase. 3-phosphoglycerate is added to a final concentration of 5 mM to start the reaction. Activity measurements are performed with a Cobas Mira Plus automated analyzer[2]. |
| References |
| Density | 1.5±0.1 g/cm3 |
|---|---|
| Boiling Point | 494.5±40.0 °C at 760 mmHg |
| Melting Point | 101-104ºC |
| Molecular Formula | C5H12O5 |
| Molecular Weight | 152.146 |
| Flash Point | 261.9±21.9 °C |
| Exact Mass | 152.068466 |
| PSA | 101.15000 |
| LogP | -3.77 |
| Vapour Pressure | 0.0±2.9 mmHg at 25°C |
| Index of Refraction | 1.571 |
| Storage condition | Store at RT. |
| Stability | Stable. Combustible. Incompatible with strong oxidizing agents. |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | VJ0800000 |
| Precursor 5 | |
|---|---|
| DownStream 10 | |


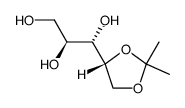
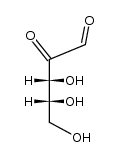
![3-(1,4-dioxaspiro[4.5]decan-3-yl)prop-2-en-1-ol structure](https://image.chemsrc.com/caspic/123/88367-50-4.png)