N-Methylparoxetine
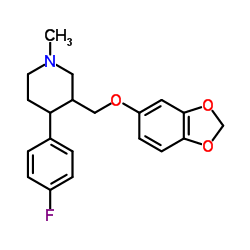
N-Methylparoxetine structure
|
Common Name | N-Methylparoxetine | ||
|---|---|---|---|---|
| CAS Number | 110429-36-2 | Molecular Weight | 343.392 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 443.7±45.0 °C at 760 mmHg | |
| Molecular Formula | C20H22FNO3 | Melting Point | 111 °C | |
| MSDS | N/A | Flash Point | 222.1±28.7 °C | |
| Symbol |


GHS06, GHS09 |
Signal Word | Danger | |
Use of N-MethylparoxetineN-methyl Paroxetine is a derivative of the selective serotonin reuptake inhibitor (SSRI) antidepressant paroxetine that inhibits [3H]paroxetine binding to rat cortical membranes. It inhibits serotonin (5-HT) uptake in rat brain synaptosomes and has been used as a precursor in the synthesis of paroxetine. |
| Name | N-Methyl Paroxetine |
|---|---|
| Synonym | More Synonyms |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 443.7±45.0 °C at 760 mmHg |
| Melting Point | 111 °C |
| Molecular Formula | C20H22FNO3 |
| Molecular Weight | 343.392 |
| Flash Point | 222.1±28.7 °C |
| Exact Mass | 343.158386 |
| PSA | 30.93000 |
| LogP | 3.91 |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.562 |
| InChIKey | MOJZPKOBKCXNKG-YJBOKZPZSA-N |
| SMILES | CN1CCC(c2ccc(F)cc2)C(COc2ccc3c(c2)OCO3)C1 |
| Storage condition | Hygroscopic, -20°C Freezer, Under Inert Atmosphere |
| Symbol |


GHS06, GHS09 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H301-H410 |
| Precautionary Statements | P273-P301 + P310-P501 |
| Hazard Codes | T,N |
| Risk Phrases | 25-50/53 |
| Safety Phrases | 45-60-61 |
| RIDADR | UN 2811 6.1 / PGIII |
| Hazard Class | 6.1 |
|
~78% 
N-Methylparoxetine CAS#:110429-36-2 |
| Literature: Segura, Mireia; Roura, Lidia; De La Torre, Rafael; Joglar, Jesus Bioorganic Chemistry, 2003 , vol. 31, # 3 p. 248 - 258 |
|
~92% 
N-Methylparoxetine CAS#:110429-36-2 |
| Literature: Wei, Peng; Kaatz, Glenn W.; Kerns, Robert J. Bioorganic and Medicinal Chemistry Letters, 2004 , vol. 14, # 12 p. 3093 - 3097 |
|
~% 
N-Methylparoxetine CAS#:110429-36-2 |
| Literature: Bioorganic Chemistry, , vol. 31, # 3 p. 248 - 258 |
|
~% 
N-Methylparoxetine CAS#:110429-36-2 |
| Literature: Synthetic Communications, , vol. 42, # 22 p. 3344 - 3360 |
|
~% 
N-Methylparoxetine CAS#:110429-36-2 |
| Literature: EP1286965 B1, ; Page 5 ; |
|
Depletion of selective serotonin reuptake inhibitors during sewage sludge composting.
Waste Manag. 29(11) , 2808-15, (2009) Sewage and sewage sludge is known to contain pharmaceuticals, and since sewage sludge is often used as fertilizer within agriculture, the reduction of the selective serotonin reuptake inhibitors (SSRI... |
|
Name: Inhibition of the Burkholderia mallei acyl-homoserine lactone synthase BmaI1
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
Target: autoinducer synthetase family protein [Burkholderia mallei ATCC 23344]
External Id: HMS1126
|
|
Name: Screen for inhibitors of RMI FANCM (MM2) intereaction
Source: 11908
Target: N/A
External Id: RMI-FANCM-MM2
|
|
Name: Chemical Probes of Kaposi's Sarcoma Herpes Virus Latent Infection
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
Target: ORF 73 [Human herpesvirus 8 type M]
External Id: HMS791
|
|
Name: A screen for compounds that inhibit the activity of LtaS in Staphylococcus aureus
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
External Id: HMS979
|
|
Name: Cell-based high throughput primary assay to identify activators of GPR151
Source: The Scripps Research Institute Molecular Screening Center
Target: RecName: Full=G-protein coupled receptor 151; AltName: Full=G-protein coupled receptor PGR7; AltName: Full=GPCR-2037; AltName: Full=Galanin receptor 4; AltName: Full=Galanin-receptor-like protein; Short=GalRL
External Id: GPR151_PHUNTER_AG_LUMI_1536_1X%ACT
|
|
Name: AlphaScreen-based biochemical high throughput primary assay to identify activators of...
Source: The Scripps Research Institute Molecular Screening Center
Target: N/A
External Id: FBW7_ACT_ALPHA_1536_1X%ACT PRUN
|
|
Name: AlphaScreen-based biochemical high throughput primary assay to identify inhibitors of...
Source: The Scripps Research Institute Molecular Screening Center
External Id: MITF_INH_Alpha_1536_1X%INH PRUN
|
|
Name: Discovering small molecule activators of G protein-gated inwardly-rectifying potassiu...
Source: 15621
Target: G protein-activated inward rectifier potassium channel 2
External Id: VANDERBILT_HTS_GIRK2_MPD
|
|
Name: Percent inhibition of EtBr efflux against Staphylococcus aureus 1199B at 50 concentra...
Source: ChEMBL
Target: Staphylococcus aureus
External Id: CHEMBL806247
|
| Paroxetine Related Compound F |
| 3-[(1,3-Benzodioxol-5-yloxy)methyl]-4-(4-fluorophenyl)-1-methylpiperidine |
| (3S,4R)-3-(1,3-benzodioxol-5-yloxymethyl)-4-(4-fluorophenyl)-1-methylpiperidine |
| MFCD03788781 |
| N-Methyl Paroxetine |
| (3S,4R)-3-[(1,3-Benzodioxol-5-yloxy)methyl]-4-(4-fluorophenyl)-1-methylpiperidine |
| Paroxetine Impurity 9 |
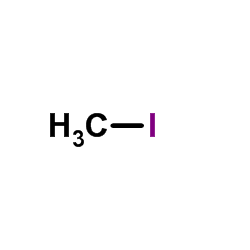
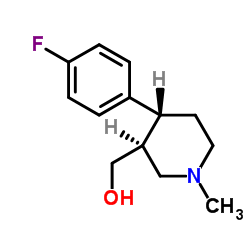

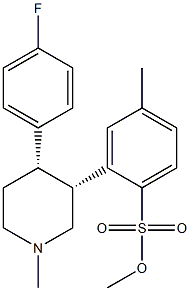
 CAS#:61869-08-7
CAS#:61869-08-7