Orotic Acid-13C,15N2 Monohydrate
Modify Date: 2024-04-03 14:58:32
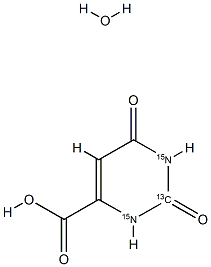
Orotic Acid-13C,15N2 Monohydrate structure
|
Common Name | Orotic Acid-13C,15N2 Monohydrate | ||
|---|---|---|---|---|
| CAS Number | 1346602-15-0 | Molecular Weight | 177.13854 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C5H6N2O5 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | N/A | |
Use of Orotic Acid-13C,15N2 MonohydrateOrotic acid-13C,15N2 (monohydrate) is the 13C and 15N labeled Orotic acid[1]. Orotic acid (6-Carboxyuracil), a precursor in biosynthesis of pyrimidine nucleotides and RNA, is released from the mitochondrial dihydroorotate dehydrogenase (DHODH) for conversion to UMP by the cytoplasmic UMP synthase enzyme. Orotic acid is a marker for measurement in routine newborn screening for urea cycle disorders. Orotic acid can induce hepatic steatosis and hepatomegaly in rats[2][3][4]. |
| Name | yxuzglgrbbhyfz-rybqtaqpsa-n |
|---|
| Description | Orotic acid-13C,15N2 (monohydrate) is the 13C and 15N labeled Orotic acid[1]. Orotic acid (6-Carboxyuracil), a precursor in biosynthesis of pyrimidine nucleotides and RNA, is released from the mitochondrial dihydroorotate dehydrogenase (DHODH) for conversion to UMP by the cytoplasmic UMP synthase enzyme. Orotic acid is a marker for measurement in routine newborn screening for urea cycle disorders. Orotic acid can induce hepatic steatosis and hepatomegaly in rats[2][3][4]. |
|---|---|
| Related Catalog | |
| In Vitro | Stable heavy isotopes of hydrogen, carbon, and other elements have been incorporated into drug molecules, largely as tracers for quantitation during the drug development process. Deuteration has gained attention because of its potential to affect the pharmacokinetic and metabolic profiles of drugs[1]. |
| References |
| Molecular Formula | C5H6N2O5 |
|---|---|
| Molecular Weight | 177.13854 |