JIB-04
Modify Date: 2025-08-25 15:51:18
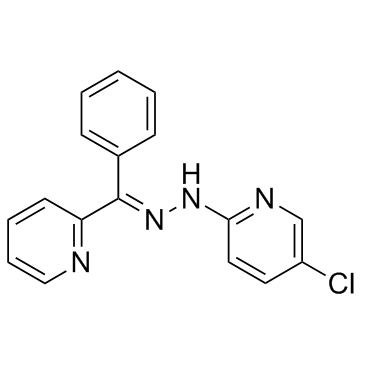
JIB-04 structure
|
Common Name | JIB-04 | ||
|---|---|---|---|---|
| CAS Number | 199596-05-9 | Molecular Weight | 308.765 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 472.9±55.0 °C at 760 mmHg | |
| Molecular Formula | C17H13ClN4 | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 239.8±31.5 °C | |
Use of JIB-04JIB-04 is a pan-selective Jumonji histone demethylase inihibitor with IC50s of 230, 340, 855, 445, 435, 1100, and 290 nM for JARID1A, JMJD2E, JMJD3, JMJD2A, JMJD2B, JMJD2C, and JMJD2D, respectively. |
| Name | 5-chloro-N-[(E)-[phenyl(pyridin-2-yl)methylidene]amino]pyridin-2-amine |
|---|---|
| Synonym | More Synonyms |
| Description | JIB-04 is a pan-selective Jumonji histone demethylase inihibitor with IC50s of 230, 340, 855, 445, 435, 1100, and 290 nM for JARID1A, JMJD2E, JMJD3, JMJD2A, JMJD2B, JMJD2C, and JMJD2D, respectively. |
|---|---|
| Related Catalog | |
| Target |
IC50: 230 nM (JARID1A), 445 nM (JMJD2A), 435 nM (JMJD2B), 1100 nM (JMJD2C), 290 nM (JMJD2D), 340 nM (JMJD2E), 855 nM (JMJD3)[1] |
| In Vitro | JIB-04 is consistently selective for cancer vs. normal cells, demonstrated by the higher sensitivity of lung and prostate cancer lines (with IC50 as low as 10 nM) compared to HBECs and PrSCs/PrECs. JIB-04 inhibits cellular Jumonji demethylase activity, and Jumonji levels affect JIB-04 action in cells[1]. JIB-04 significantly inhibits the proliferation of GB cell lines and stem-enriched cultures. JIB-04 exerts its maximal inhibitory activity against KDM5A, and modulates the expression of genes involved in the control of cancer cell growth and leads to hypermethylation of H3K4. Furthermore, JIB-04 (2500 nM) activates the autophagy and apoptotic pathways and inactivates PI3K. JIB-04 also cooperates with TMZ in killing GB cells[2]. |
| In Vivo | JIB-04 results in a significant reduction in cancer-induced death rates in mice, prolonging survival[1]. JIB-04 (60, 40 and 20 mg/kg, i.p.) reaches bioactive concentration in the brain of the mice. The orthotopic GB xenograft model shows a trend toward longer survival in JIB-04-treated mice with an Hazard Ratio of 0.5[2]. |
| Cell Assay | For cell viability assays, cells are plated at 1500-3000 cells/well in 96 well plates and treated the next day with increasing doses of compound over 4 days and their viability assessed by standard MTS assays using Promega’s Cell Titer or Cell Titer-Glo reagents. Absorbance at 490 nm and 650 nm or luminescence is measured by a Spectra Max or a FlouroStar Omega plate reader. Data are normalized to the untreated controls (100% viability). Each cell line is tested in 2-5 independent assays, each containing 4-8 replicates. IC50 values are calculated using DIVISA, a high-throughput software, developed in house, for storing and analyzing drug sensitivity assays. Dose-response curves are plotted using a non-linear regression model and IC50s are determined from the fitted curves. The average IC50 derived from 2-5 independent assays, each containing 4-8 replicates is reported. |
| Animal Admin | For xenografts, 4-6 week old female nude mice are housed under standard conditions in a clean facility at UTSW. Two million H358 cells or five million A549 cells are injected subcutaneously and allowed to grow for 2-3 weeks with monitoring. When tumors reach appr 200 mm3, therapy is started in weight and tumor volume matched pairs (n=7 for each treatment group for each cell line). Drug or vehicle is administered by inter-peritoneal injection in 10% DMSO 90% sesame oil 2 to 3 times weekly for 5 weeks at 110 mg/kg to all mice harboring H358 xenografts or 3 times per week by gavage in 12.5% Cremophor EL, 12.5% DMSO as an aqueous suspension at 55 mg/kg to mice harboring A549 xenografts. Tumor volumes are monitored twice weekly by caliper measurements. Animals are weighed and observed during the five weeks of treatment. At the end point, mice are euthanized by CO2 asphyxiation and cervical dislocation, and blood, tumors and major organs collected and weighed. Paired, unequal variance, one-tailed t-tests are performed across treatment groups using Excel software. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 472.9±55.0 °C at 760 mmHg |
| Molecular Formula | C17H13ClN4 |
| Molecular Weight | 308.765 |
| Flash Point | 239.8±31.5 °C |
| Exact Mass | 308.082886 |
| PSA | 50.17000 |
| LogP | 3.92 |
| Appearance of Characters | white to beige |
| Vapour Pressure | 0.0±1.2 mmHg at 25°C |
| Index of Refraction | 1.644 |
| InChIKey | YHHFKWKMXWRVTJ-UHFFFAOYSA-N |
| SMILES | Clc1ccc(NN=C(c2ccccc2)c2ccccn2)nc1 |
| Storage condition | 2-8°C |
| Water Solubility | DMSO: soluble10mg/mL, clear |
| RIDADR | NONH for all modes of transport |
|---|
|
Name: Inhibition of KDM5B (unknown origin)
Source: ChEMBL
Target: Lysine-specific demethylase 5B
External Id: CHEMBL5349606
|
|
Name: Inhibition of N-terminal His-tagged recombinant human TET2 catalytic domain LCIdel mu...
Source: ChEMBL
Target: N/A
External Id: CHEMBL5337141
|
|
Name: Antiproliferative activity against human A549 cells assessed as reduction in cell via...
Source: ChEMBL
Target: A549
External Id: CHEMBL5237420
|
|
Name: Inhibition of His10-FLAG tagged human TET2 catalytic domain (Q969 to I2002 residues) ...
Source: ChEMBL
Target: Methylcytosine dioxygenase TET2
External Id: CHEMBL5337139
|
|
Name: Inhibition of human TET3 catalytic domain (E824 to I1795 residues) expressed in mamma...
Source: ChEMBL
Target: Methylcytosine dioxygenase TET3
External Id: CHEMBL5337140
|
|
Name: Antiviral activity determined as inhibition of SARS-CoV-2 induced cytotoxicity of VER...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL4513082
|
|
Name: Inhibition of N-terminal 3xFlag-tagged human TET1 catalytic domain (E1418 to V2136 re...
Source: ChEMBL
Target: Methylcytosine dioxygenase TET1
External Id: CHEMBL5337138
|
|
Name: Antiviral activity determined as inhibition of SARS-CoV-2 induced cytotoxicity of Cac...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL4303805
|
|
Name: SARS-CoV-2 3CL-Pro protease inhibition percentage at 20µM by FRET kind of response f...
Source: ChEMBL
Target: Replicase polyprotein 1ab
External Id: CHEMBL4495582
|
|
Name: Enzymatic assay of human HDAC6 with commercial peptide substrate
Source: ChEMBL
Target: Histone deacetylase 6
External Id: CHEMBL4808149
|
Total 23, Current Page 1 of 3
1
2
3
| jib-04 |
| 5-Chloro-2-{(2E)-2-[phenyl(2-pyridinyl)methylene]hydrazino}pyridine |
| fd5031 |