tesaglitazar
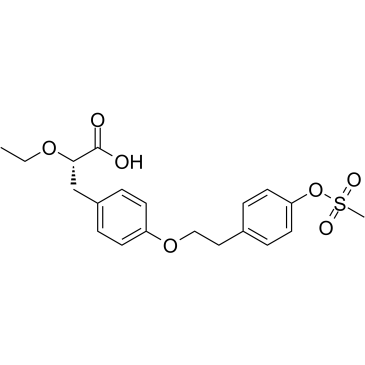
tesaglitazar structure
|
Common Name | tesaglitazar | ||
|---|---|---|---|---|
| CAS Number | 251565-85-2 | Molecular Weight | 408.465 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 611.3±55.0 °C at 760 mmHg | |
| Molecular Formula | C20H24O7S | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 323.5±31.5 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of tesaglitazarTesaglitazar is a dual peroxisome proliferator-activated receptor (PPAR) alpha/gamma agonist that is more potent on PPARγ than on PPARα, with EC50s of 13.4 μM and 3.6 μM for rat PPARα and human PPARα, respectively, and approximately 0.2 μM for both rat and human PPARγ. Tesaglitazar induces interstitial mesenchymal cell DNA synthesis and fibrosarcomas in subcutaneous tissues in rats[1]. |
| Name | (2S)-2-ethoxy-3-[4-[2-(4-methylsulfonyloxyphenyl)ethoxy]phenyl]propanoic acid |
|---|---|
| Synonym | More Synonyms |
| Description | Tesaglitazar is a dual peroxisome proliferator-activated receptor (PPAR) alpha/gamma agonist that is more potent on PPARγ than on PPARα, with EC50s of 13.4 μM and 3.6 μM for rat PPARα and human PPARα, respectively, and approximately 0.2 μM for both rat and human PPARγ. Tesaglitazar induces interstitial mesenchymal cell DNA synthesis and fibrosarcomas in subcutaneous tissues in rats[1]. |
|---|---|
| Related Catalog | |
| Target |
EC50: 13.4 μM (rat PPARα), 3.6 μM (human PPARα), 0.2 μM (PPARγ)[1] |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 611.3±55.0 °C at 760 mmHg |
| Molecular Formula | C20H24O7S |
| Molecular Weight | 408.465 |
| Flash Point | 323.5±31.5 °C |
| Exact Mass | 408.124268 |
| PSA | 107.51000 |
| LogP | 2.64 |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.566 |
| InChIKey | CXGTZJYQWSUFET-IBGZPJMESA-N |
| SMILES | CCOC(Cc1ccc(OCCc2ccc(OS(C)(=O)=O)cc2)cc1)C(=O)O |
| Storage condition | 2-8°C |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302 |
| Precautionary Statements | P301 + P312 + P330 |
| RIDADR | NONH for all modes of transport |
| HS Code | 2918990090 |
|
~80% 
tesaglitazar CAS#:251565-85-2 |
| Literature: AstraZeneca AB Patent: US6258850 B1, 2001 ; |
|
~% 
tesaglitazar CAS#:251565-85-2 |
| Literature: WO2003/82812 A2, ; Page/Page column 6 ; WO 03/082812 A2 |
| Precursor 4 | |
|---|---|
| DownStream 0 | |
| HS Code | 2918990090 |
|---|---|
| Summary | 2918990090. other carboxylic acids with additional oxygen function and their anhydrides, halides, peroxides and peroxyacids; their halogenated, sulphonated, nitrated or nitrosated derivatives. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Name: This assay (PMID: 37558952) monitors the impact of a chemical compound on general aut...
Source: ChEMBL
Target: TERT-RPE1
External Id: CHEMBL5724699
|
|
Name: Antihyperglycemic activity in db/db mouse assessed as reduction in glucose level at 3...
Source: ChEMBL
Target: Mus musculus
External Id: CHEMBL1815141
|
|
Name: Thermal Shift Assay. Domain: start/stop: M1-L298
Source: ChEMBL
Target: Cyclin-dependent kinase 2
External Id: CHEMBL5062802
|
|
Name: Agonist activity at GAL4N fused human PPARgamma LBD expressed in HEK293 cells co-expr...
Source: ChEMBL
Target: Peroxisome proliferator-activated receptor gamma
External Id: CHEMBL4022336
|
|
Name: Agonist activity at GAL4N fused human PPARalpha LBD expressed in HEK293 cells co-expr...
Source: ChEMBL
Target: Peroxisome proliferator-activated receptor alpha
External Id: CHEMBL4022334
|
|
Name: Agonist activity at GAL4N fused human PPARdelta LBD expressed in HEK293 cells co-expr...
Source: ChEMBL
Target: Peroxisome proliferator-activated receptor delta
External Id: CHEMBL4022346
|
|
Name: Agonist activity at GAL4-fused PPARalpha assessed as transcriptional activity by cell...
Source: ChEMBL
Target: Peroxisome proliferator-activated receptor alpha
External Id: CHEMBL2216559
|
|
Name: Inhibition of PPARalpha at 50 uM
Source: ChEMBL
Target: Peroxisome proliferator-activated receptor alpha
External Id: CHEMBL949956
|
|
Name: Inhibition of PPARgamma at 1 uM
Source: ChEMBL
Target: Peroxisome proliferator-activated receptor gamma
External Id: CHEMBL949957
|
|
Name: Agonist activity at GAL4-fused PPARgamma assessed as transcriptional activity by cell...
Source: ChEMBL
Target: Peroxisome proliferator-activated receptor gamma
External Id: CHEMBL2216558
|
| [14C]-Tesaglitazar |
| (2S)-2-Ethoxy-3-[4-(2-{4-[(methylsulfonyl)oxy]phenyl}ethoxy)phenyl]propanoic acid |
| Galida |
| tesaglitazar |
| (S)-2-ethoxy-3-[4-(2-{4-methanesulfonyloxy-phenyl}ethoxy)phenyl]propanoic acid |
| AZ-242 |
| (S)-2-Ethoxy-3-{4-[2-(4-methanesulfonyloxyphenyl)ethoxy]phenyl}propionic acid |
| (S)-2-ethoxy-3-[4-(2-{4-methanesulphonyloxyphenyl}ethoxy)phenyl]propanoic acid |
| (2S)-2-ethoxy-3-[4-(2-{4-[(methylsuIfonyl)oxy]phenyl}ethoxy)phenyl]propanoic acid |
| AZ2 |
| (2S)-2-Ethoxy-3-[4-[2-[4-[(methylsulfonyl)oxy]phenyl]ethoxy]phenyl]propanoic acid |
| (S)-2-ethoxy-3-(4-(2-(4-methylsulphonyloxyphenyl)ethoxy)phenyl)propanoic acid |
![(S)-2-ethoxy-N-(2-hydroxy-(R)-1-phenylethyl)-3-[4-(2-{4-methanesulfonyloxyphenyl}-ethoxy)phenyl]propanoic amide structure](https://image.chemsrc.com/caspic/353/251565-89-6.png)
![ethyl (2S)-2-ethoxy-3-[4-(2-{4-[(methylsulfonyl)oxy]phenyl}ethoxy)phenyl]propanoate structure](https://image.chemsrc.com/caspic/450/251565-91-0.png)