Apiin
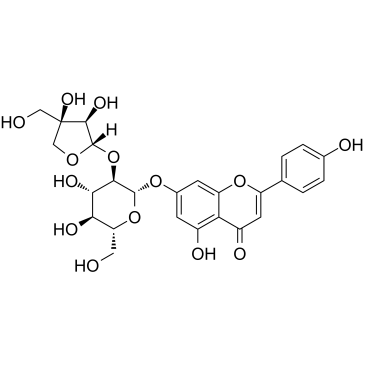
Apiin structure
|
Common Name | Apiin | ||
|---|---|---|---|---|
| CAS Number | 26544-34-3 | Molecular Weight | 564.492 | |
| Density | 1.7±0.1 g/cm3 | Boiling Point | 942.2±65.0 °C at 760 mmHg | |
| Molecular Formula | C26H28O14 | Melting Point | 230ºC (dec.) | |
| MSDS | Chinese USA | Flash Point | 316.7±27.8 °C | |
Use of ApiinApiin, a major constituent of Apium graveolens leaves with anti-inflammatory properties. Apiin shows significant inhibitory activity on nitrite (NO) production (IC50 = 0.08 mg/mL) in-vitro and iNOS expression (IC50 = 0.049 mg/ mL) in LPS-activated J774.A1 cells[1]. |
| Name | Apiin |
|---|---|
| Synonym | More Synonyms |
| Description | Apiin, a major constituent of Apium graveolens leaves with anti-inflammatory properties. Apiin shows significant inhibitory activity on nitrite (NO) production (IC50 = 0.08 mg/mL) in-vitro and iNOS expression (IC50 = 0.049 mg/ mL) in LPS-activated J774.A1 cells[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.7±0.1 g/cm3 |
|---|---|
| Boiling Point | 942.2±65.0 °C at 760 mmHg |
| Melting Point | 230ºC (dec.) |
| Molecular Formula | C26H28O14 |
| Molecular Weight | 564.492 |
| Flash Point | 316.7±27.8 °C |
| Exact Mass | 564.147888 |
| PSA | 228.97000 |
| LogP | 0.74 |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.744 |
| InChIKey | NTDLXWMIWOECHG-YRCFQSNFSA-N |
| SMILES | O=c1cc(-c2ccc(O)cc2)oc2cc(OC3OC(CO)C(O)C(O)C3OC3OCC(O)(CO)C3O)cc(O)c12 |
| Storage condition | 2-8C |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |
| WGK Germany | 1 |
| HS Code | 29389090 |
| Precursor 0 | |
|---|---|
| DownStream 3 | |
|
Evaluation of detection sensitivity in comprehensive two-dimensional liquid chromatography separations of an active pharmaceutical ingredient and its degradants.
Anal. Bioanal. Chem 407(1) , 265-77, (2015) In this paper, we describe the findings of a study aimed at assessing the detection sensitivity of comprehensive two-dimensional high-performance liquid chromatography (LCxLC) separation of a degraded... |
|
|
Bioavailability of apigenin from apiin-rich parsley in humans.
Ann. Nutr. Metab. 50(3) , 167-72, (2006) Absorption and excretion of apigenin after the ingestion of apiin-rich food, i.e. parsley, was tested.Eleven healthy subjects (5 women, 6 men) in the age range of 23-41 years and with an average body ... |
|
|
[Studies on the chemical constituents of the aerial parts of Seseli mairei].
Zhong Yao Cai 30(1) , 42-4, (2007) To study the chemical constituents of the aerial parts of Seseli mairei Wolf.The chemical constituents have been separated with manifold chromatography methods, and their structures were determined hy... |
|
Name: Primary cell-based high-throughput screening assay for identification of compounds th...
Source: Johns Hopkins Ion Channel Center
Target: regulator of G-protein signaling 4 isoform 2 [Homo sapiens]
External Id: JHICC_RGS_Act_HTS
|
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: HepG2 viability counterscreen (readout 2: ATP content) against the NCATS DSHEA and TC...
Source: NCGC
Target: N/A
External Id: DSHEA-v1-HepG2-viability-CTG-ATP
|
|
Name: ERK5 transcriptional activity HTS
Source: 24565
Target: N/A
External Id: ERK5 transcriptional activity-HTS
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: uHTS identification of small molecule activators of the adaptive arm of the Unfolded ...
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: BCCG-A405-UPR-XBP1-PrimaryAgonist-Assay
|
|
Name: High throughput fluorescence intensity-based biochemical assay to screen for small mo...
Source: University of Pittsburgh Molecular Library Screening Center
Target: furin (paired basic amino acid cleaving enzyme), isoform CRA_a [Homo sapiens]
External Id: MH080376 Biochemical HTS for Inhibitors of the Proprotein Convertase Furin.
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
|
Name: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfect...
Source: Broad Institute
Target: N/A
External Id: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfected HEK293 cells Inhibition - 7011-01_Antagonist_SinglePoint_HTS_Activity
|
| APIIN |
| Apioside |
| APIGENIN-7-APIOGLUCOSIDE |
| 5-Hydroxy-2-(4-hydroxyphenyl)-4-oxo-4H-chromen-7-yl 4-O-[(3R,4R)-3,4-dihydroxy-4-(hydroxymethyl)tetrahydrofuran-2-yl]-β-D-glucopyranoside |
| apigenin-7-O-apiosyl(1->2)glucoside |
| apigenin 7-O-apiosylglucoside |
| 4',5,7-Trihydroxyflavone-7-apiosylglucoside |
| 5-Hydroxy-2-(4-hydroxyphenyl)-4-oxo-4H-chromen-7-yl 4-O-[(3R,4R)-3,4-dihydroxy-4-(hydroxymethyl)tetrahydro-2-furanyl]-β-D-glucopyranoside |
| 7-(2-APIOSYLGLUCOSYL)APIGENIN |
| Apigenin-7-(2-O-apiosylglucoside) |
| 7-[(2-O-D-Apio-b-D-furanosyl-b-D-glucopyranosyl)oxy]-5-hydroxy-2-(4-hydroxyphenyl)-4H-1-benzopyran-4-one |
| Apigenin-7-apiosylglucoside |
| EINECS 247-780-0 |
 CAS#:2280-44-6
CAS#:2280-44-6 CAS#:520-36-5
CAS#:520-36-5 CAS#:578-74-5
CAS#:578-74-5
