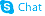5-chlorosalicylic acid
5-chlorosalicylic acid structure
|
Common Name | 5-chlorosalicylic acid | ||
|---|---|---|---|---|
| CAS Number | 321-14-2 | Molecular Weight | 172.566 | |
| Density | 1.5±0.1 g/cm3 | Boiling Point | 320.5±27.0 °C at 760 mmHg | |
| Molecular Formula | C7H5ClO3 | Melting Point | 171-172 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 147.6±23.7 °C | |
| Symbol |

GHS06 |
Signal Word | Danger | |
| Name | 5-chlorosalicylic acid |
|---|---|
| Synonym | More Synonyms |
| Density | 1.5±0.1 g/cm3 |
|---|---|
| Boiling Point | 320.5±27.0 °C at 760 mmHg |
| Melting Point | 171-172 °C(lit.) |
| Molecular Formula | C7H5ClO3 |
| Molecular Weight | 172.566 |
| Flash Point | 147.6±23.7 °C |
| Exact Mass | 171.992722 |
| PSA | 57.53000 |
| LogP | 3.42 |
| Vapour Pressure | 0.0±0.7 mmHg at 25°C |
| Index of Refraction | 1.630 |
| Water Solubility | 1 g/L (20 ºC) |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H301-H315-H319 |
| Precautionary Statements | P301 + P310-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Faceshields;Gloves |
| Hazard Codes | Xn:Harmful |
| Risk Phrases | R22;R36/37/38 |
| Safety Phrases | S26-S36-S37/39 |
| RIDADR | UN 2811 6.1/PG 3 |
| WGK Germany | 3 |
| RTECS | VO2080000 |
| Packaging Group | III |
| Hazard Class | 6.1 |
| HS Code | 29182990 |
| Precursor 9 | |
|---|---|
| DownStream 10 | |
| HS Code | 2918290000 |
|---|---|
| Summary | HS: 2918290000 other carboxylic acids with phenol function but without other oxygen function, their anhydrides, halides, peroxides, peroxyacids and their derivatives Tax rebate rate:9.0% Supervision conditions:AB(certificate of inspection for goods inward,certificate of inspection for goods outward) VAT:17.0% MFN tariff:6.5% General tariff:30.0% |
|
Predicting human serum albumin affinity of interleukin-8 (CXCL8) inhibitors by 3D-QSPR approach.
J. Med. Chem. 48 , 2469-79, (2005) A novel class of 2-(R)-phenylpropionamides has been recently reported to inhibit in vitro and in vivo interleukin-8 (CXCL8)-induced biological activities. These CXCL8 inhibitors are derivatives of phe... |
|
|
Structure-activity relationship of salicylic acid derivatives on inhibition of TNF-α dependent NFκB activity: Implication on anti-inflammatory effect of N-(5-chlorosalicyloyl)phenethylamine against experimental colitis.
Eur. J. Med. Chem. 48 , 36-44, (2012) To develop a more potent NFκB inhibitor from salicylic acid which is known to inhibit activity of NFκB, a transcription factor regulating genes involved in immunity, inflammation and tumorigenesis, de... |
|
|
In vitro inhibition of salicylic acid derivatives on human cytosolic carbonic anhydrase isozymes I and II.
Bioorg. Med. Chem. 16 , 9101-5, (2008) The inhibition of two human cytosolic carbonic anhydrase (hCA, EC 4.2.1.1) isozymes, hCA I and II, with a series of salicylic acid derivatives was investigated by using the esterase method with 4-nitr... |
| 5-Chlorosalicyclic acid |
| 5-chloro-2-hydroxy-benzoic acid |
| Benzoic acid, 5-chloro-2-hydroxy- |
| 5-chlorosalicylic acid |
| EINECS 206-283-9 |
| Salicylic acid,5-chloro |
| 2-Hydroxy-5-chlorobenzoic acid |
| tert-butyl carbazate |
| 5-Chloro-2-hydroxybenzoic acid |
| MFCD00002457 |
| 5 CSA |
| Benzoic acid,5-chloro-2-hydroxy |
| 5-chloro-salicylic acid |
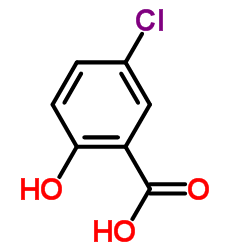
 CAS#:535-80-8
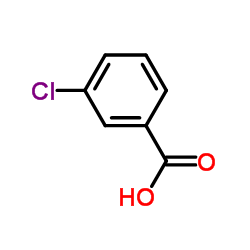
CAS#:535-80-8 CAS#:50-79-3
CAS#:50-79-3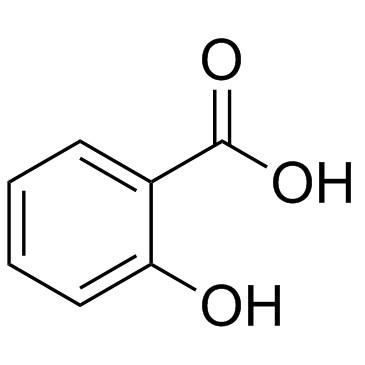 CAS#:69-72-7
CAS#:69-72-7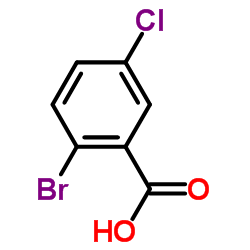 CAS#:21739-93-5
CAS#:21739-93-5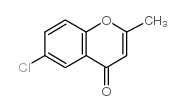 CAS#:69693-00-1
CAS#:69693-00-1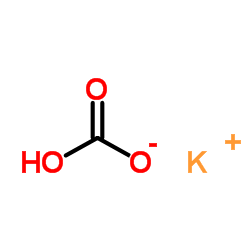 CAS#:298-14-6
CAS#:298-14-6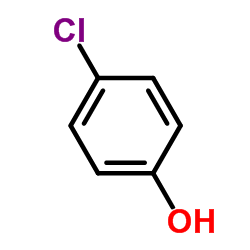 CAS#:106-48-9
CAS#:106-48-9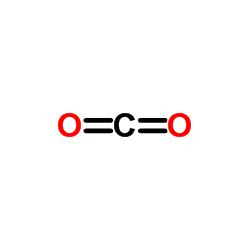 CAS#:124-38-9
CAS#:124-38-9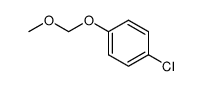 CAS#:826-26-6
CAS#:826-26-6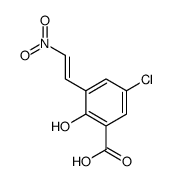 CAS#:111870-28-1
CAS#:111870-28-1![(5E)-3-chloro-N-(4-chlorophenyl)-5-[(4-fluoroanilino)methylidene]-6-oxocyclohexa-1,3-diene-1-carboxamide structure](https://www.chemsrc.com/caspic/352/111870-33-8.png) CAS#:111870-33-8
CAS#:111870-33-8![5-chloro-N-(4-chlorophenyl)-2-hydroxy-3-[(E)-2-nitroethenyl]benzamide structure](https://www.chemsrc.com/caspic/428/111870-31-6.png) CAS#:111870-31-6
CAS#:111870-31-6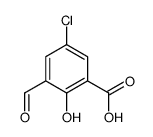 CAS#:111870-27-0
CAS#:111870-27-0![(5E)-3-chloro-5-[(4-fluoroanilino)methylidene]-N-(4-fluorophenyl)-6-oxocyclohexa-1,3-diene-1-carboxamide structure](https://www.chemsrc.com/caspic/314/111870-34-9.png) CAS#:111870-34-9
CAS#:111870-34-9![5-chloro-2-hydroxy-3-[(E)-2-nitroethenyl]-N-phenylbenzamide structure](https://www.chemsrc.com/caspic/466/111870-30-5.png) CAS#:111870-30-5
CAS#:111870-30-5![5-chloro-2-hydroxy-3-[(E)-2-nitroethenyl]benzoyl chloride structure](https://www.chemsrc.com/caspic/321/111870-29-2.png) CAS#:111870-29-2
CAS#:111870-29-2 CAS#:3261-05-0
CAS#:3261-05-0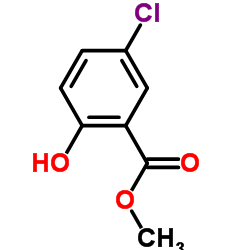 CAS#:4068-78-4
CAS#:4068-78-4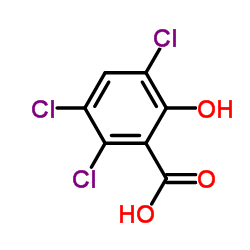 CAS#:40932-60-3
CAS#:40932-60-3