phosphoramidon
Modify Date: 2025-08-24 06:49:46
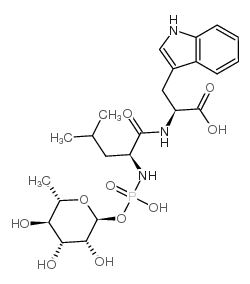
phosphoramidon structure
|
Common Name | phosphoramidon | ||
|---|---|---|---|---|
| CAS Number | 36357-77-4 | Molecular Weight | 543.50400 | |
| Density | 1.48 g/cm3 | Boiling Point | N/A | |
| Molecular Formula | C23H34N3O10P | Melting Point | N/A | |
| MSDS | N/A | Flash Point | N/A | |
Use of phosphoramidonPhosphoramidon, a microbial metabolite, is a specific metalloprotease thermolysin inhibitor with an IC50 of 0.4 μg/mL. Phosphoramidon also inhibits endothelin-converting enzyme (ECE), neutral endopeptidase (NEP), and angiotensin-converting enzyme (ACE) with IC50 values of 3.5, 0.034, and 78 μM, respectively[1][2][3]. |
| Name | phosphoramidon |
|---|---|
| Synonym | More Synonyms |
| Description | Phosphoramidon, a microbial metabolite, is a specific metalloprotease thermolysin inhibitor with an IC50 of 0.4 μg/mL. Phosphoramidon also inhibits endothelin-converting enzyme (ECE), neutral endopeptidase (NEP), and angiotensin-converting enzyme (ACE) with IC50 values of 3.5, 0.034, and 78 μM, respectively[1][2][3]. |
|---|---|
| Related Catalog | |
| Target |
Microbial Metabolite |
| In Vitro | Phosphoramidon (1-500 μM; 30 min) inhibits ET-converting enzyme (ECE) activity in a dose-dependent manner in solubilized rabbit lung membranes[5]. |
| In Vivo | Phosphoramidon (0.25 mg/kg per min; i.v.) suppresses the hypertensive effect of big endothelin-1 in rats[4]. Phosphoramidon (1-30 mg/kg; i.v.; once) blocks the pressor activity of porcine big endothelin-1-(1-39) in rats[5]. Animal Model: Male Sprague-Dawley rats[4] Dosage: 0.25 mg/kg per min Administration: Intravenous injection Result: Markedly suppressed the hypertensive effect of big endothelin-1. |
| Density | 1.48 g/cm3 |
|---|---|
| Molecular Formula | C23H34N3O10P |
| Molecular Weight | 543.50400 |
| Exact Mass | 543.19800 |
| PSA | 220.48000 |
| LogP | 1.01040 |
| Index of Refraction | 1.631 |
| InChIKey | ZPHBZEQOLSRPAK-XLCYBJAPSA-N |
| SMILES | CC(C)CC(NP(=O)(O)OC1OC(C)C(O)C(O)C1O)C(=O)NC(Cc1c[nH]c2ccccc12)C(=O)O |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Safety Phrases | 22-24/25 |
|---|
| Precursor 0 | |
|---|---|
| DownStream 2 | |
|
Name: Inhibition of collagenase type 4 after 30 mins by fluorescence plate reader
Source: ChEMBL
Target: N/A
External Id: CHEMBL1066257
|
|
Name: Inhibition of human somatic ECE1
Source: ChEMBL
Target: Endothelin-converting enzyme 1
External Id: CHEMBL1045249
|
|
Name: In vitro inhibition to recombinant human endothelin converting enzyme
Source: ChEMBL
Target: Endothelin-converting enzyme 1
External Id: CHEMBL675442
|
|
Name: Inhibition of Pseudomonas aeruginosa LasB elastase using aminobenzoyl-Ala-Gly-Leu-Ala...
Source: ChEMBL
Target: Elastase
External Id: CHEMBL5257279
|
|
Name: Experimentally measured binding affinity data (Ki) for protein-ligand complexes deriv...
Source: Shanghai Institute of Organic Chemistry
Target: N/A
External Id: PDBbind-Ki for protein-ligand complexes
|
|
Name: Antiviral activity determined as inhibition of SARS-CoV-2 induced cytotoxicity of VER...
Source: ChEMBL
Target: Severe acute respiratory syndrome coronavirus 2
External Id: CHEMBL4513082
|
|
Name: Ratio of inhibitory activities against endothelin converting enzyme-1(ECE-1) and neut...
Source: ChEMBL
Target: N/A
External Id: CHEMBL678352
|
|
Name: Inhibition of Endothelin-converting enzyme 1 activity at a concentration of 1 uM
Source: ChEMBL
Target: Endothelin-converting enzyme 1
External Id: CHEMBL878272
|
Total 20, Current Page 1 of 2
1
2
| PHOSPHORAMIDON SODIUM SALT |
| yptophan |
| phophoramidon |
| eucyl) |
| PHOSPHORAMIDON |
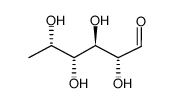 CAS#:7658-10-8
CAS#:7658-10-8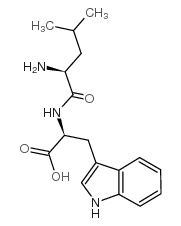 CAS#:5156-22-9
CAS#:5156-22-9
