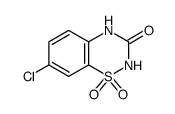
diazoxide
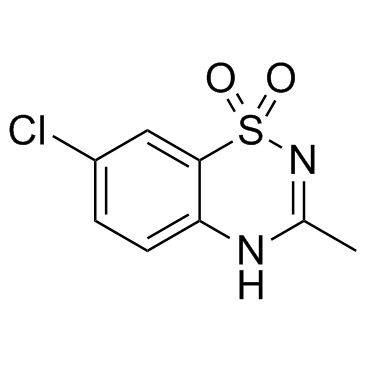
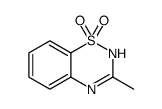
diazoxide structure
|
Common Name | diazoxide | ||
|---|---|---|---|---|
| CAS Number | 364-98-7 | Molecular Weight | 230.671 | |
| Density | 1.6±0.1 g/cm3 | Boiling Point | 414.8±47.0 °C at 760 mmHg | |
| Molecular Formula | C8H7ClN2O2S | Melting Point | >310°C | |
| MSDS | Chinese USA | Flash Point | 204.6±29.3 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of diazoxideDiazoxide is an ATP-sensitive potassium channel activator ; can be used to treat hyperinsulinism. |
| Name | diazoxide |
|---|---|
| Synonym | More Synonyms |
| Description | Diazoxide is an ATP-sensitive potassium channel activator ; can be used to treat hyperinsulinism. |
|---|---|
| Related Catalog | |
| In Vitro | Diazoxide has a number of physiological effects, including lowering the blood pressure and rectifying hypoglycemia. Diazoxide has powerful protective properties against cardiac ischemia[1].Diazoxide could protect NSC-34 neurons against the main sources of neurodegenerative damage. Diazoxide increases Nrf2 nuclear translocation in NSC-34 motoneurons and prevents endogenous oxidative damage[2]. |
| In Vivo | Diazoxide attenuates postresuscitation brain injury, protects mitochondrial function, inhibits brain cell apoptosis, and activates the PKC pathway by opening mitoKATP channels[3]. Treatment with diazoxide in wild-type mice decreases intraocular pressure (IOP) by 21.5±3.2% with an absolute IOP reduction of 3.9 ± 0.6 mm Hg[4]. |
| Cell Assay | Diazoxide is dissolved in DMSO to prepare 50 mM stock solution. NSC-34 cells are allowed to differentiate for 8 weeks under reduced serum conditions and then seeded in 24-well plates. Glutamate is dissolved in culture medium and added to cultures at concentration of 10 μM for 24 h. Cell treatment with 100 µM diazoxide starts 2 h before glutamate exposure. Cell viability is measured by the MTT assay[2]. |
| Animal Admin | Rats: Adult male Sprague-Dawley rats with induced cerebral ischemia (n=10 per group) receive an intraperitoneal injection of 0.1% DMSO (1 mL; vehicle group), diazoxide (10 mg/kg; DZ group), or diazoxide (10 mg/kg) plus 5-hydroxydecanoate (5 mg/kg; DZ + 5-HD group) 30 min after CPR. The control group (sham group, n=5) undergoes sham operation, without cardiac arrest. Mitochondrial respiratory control rate (RCR) is determined. Brain cell apoptosis is assessed using TUNEL staining. Expression of Bcl-2, Bax, and protein kinase C epsilon (PKCε) in the cerebral cortex is determined by Western blotting and immunohistochemistry[3]. Mouse: Diazoxide is prepared by diluting a 100 mM stock solution in 10% polyethoxylated castor oil in PBS. In C57BL/6 wild-type and Kir6.2(−/−) mice, a 5 μL drop of 5 mM diazoxide is topically administered to one eye of each mouse while the fellow control eye received vehicle (DMSO and 10% polyethoxylated castor oil in the same proportion as the treated eye). IOP is measured daily at 1 hour, 4 hours, and 23 hours following treatment. Treatment with diazoxide and vehicle is continued daily for 14 consecutive days[4]. |
| References |
| Density | 1.6±0.1 g/cm3 |
|---|---|
| Boiling Point | 414.8±47.0 °C at 760 mmHg |
| Melting Point | >310°C |
| Molecular Formula | C8H7ClN2O2S |
| Molecular Weight | 230.671 |
| Flash Point | 204.6±29.3 °C |
| Exact Mass | 229.991669 |
| PSA | 66.91000 |
| LogP | 1.08 |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.692 |
| Storage condition | Store at RT |
| Water Solubility | 0.1 M NaOH: soluble |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn: Harmful; |
| Risk Phrases | R22;R36/37/38 |
| Safety Phrases | S22;S26;S36 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | DK8185000 |
| HS Code | 2933990090 |
|
~90% 
diazoxide CAS#:364-98-7 |
| Literature: Khelili, Smail; Faury, Gilles; Nicolle, Edwige; Verdetti, Jean; Leclerc, Gerard Medicinal Chemistry Research, 2003 , vol. 12, # 9 p. 457 - 470 |
|
~% 
diazoxide CAS#:364-98-7 |
| Literature: Medicinal Chemistry Research, , vol. 12, # 9 p. 457 - 470 |
|
~% 
diazoxide CAS#:364-98-7 |
| Literature: Medicinal Chemistry Research, , vol. 12, # 9 p. 457 - 470 |
|
~% 
diazoxide CAS#:364-98-7 |
| Literature: Medicinal Chemistry Research, , vol. 12, # 9 p. 457 - 470 |
|
~% 
diazoxide CAS#:364-98-7 |
| Literature: Medicinal Chemistry Research, , vol. 12, # 9 p. 457 - 470 |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Magnesium isoglycyrrhizinate protects hepatic L02 cells from ischemia/reperfusion induced injury.
Int. J. Clin. Exp. Pathol. 7(8) , 4755-64, (2014) Human liver ischemia/reperfusion injury (IRI) is a common and major clinical problem complicating liver surgery and transplantation. The pathogenesis underlying IRI is complex, involving a series of s... |
|
|
Accelerated recovery of mitochondrial membrane potential by GSK-3β inactivation affords cardiomyocytes protection from oxidant-induced necrosis.
PLoS ONE 9(11) , e112529, (2014) Loss of mitochondrial membrane potential (ΔΨm) is known to be closely linked to cell death by various insults. However, whether acceleration of the ΔΨm recovery process prevents cell necrosis remains ... |
|
|
ATP-sensitive potassium (K(ATP)) channel openers diazoxide and nicorandil lower intraocular pressure in vivo.
Invest. Ophthalmol. Vis. Sci. 54(7) , 4892-9, (2013) To evaluate the expression of ATP-sensitive potassium (K(ATP)) channel subunits and study the effect of K(ATP) channel openers diazoxide and nicorandil on intraocular pressure (IOP) in an in vivo mous... |
| diazoxidum [INN_la] |
| 7-chloro-3-methyl-4H-1λ<sup>6</sup>,2,4-benzothiadiazine 1,1-dioxide |
| Hypertonalum |
| Diazoxido |
| 7-chloro-3-methyl-4H-1,2,4-benzothiadiazine 1,1-dioxide |
| Sodium dodecyl sulphate solution |
| EINECS 206-668-1 |
| Mutabase |
| Proglycem |
| Dizoxide |
| Diazossido |
| MFCD00078578 |
| diazoxide |
| Eudemine |
| Proglicem |
| 2H-1,2,4-Benzothiadiazine, 7-chloro-3-methyl-, 1,1-dioxide |
| 7-chloro-3-methyl-1,2,4-benzothiadiazine-1,1-dioxide |
| Hyperstat |
| 7-Chloro-3-methyl-2H-1,2,4-benzothiadiazine 1,1-dioxide |
| 7-chloro-3-méthyl-2H-1,2,4-benzothiadiazine-1,1-dioxyde |
| 3-Methyl-7-chloro-1,2,4-benzothiadiazine 1,1-dioxide |