tetryl
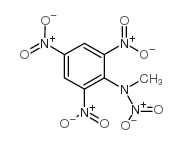
tetryl structure
|
Common Name | tetryl | ||
|---|---|---|---|---|
| CAS Number | 479-45-8 | Molecular Weight | 287.14300 | |
| Density | 1.803g/cm3 | Boiling Point | 503.7ºC at 760mmHg | |
| Molecular Formula | C7H5N5O8 | Melting Point | 130ºC | |
| MSDS | N/A | Flash Point | 258.4ºC | |
| Symbol |


GHS02, GHS07 |
Signal Word | Danger | |
| Name | N-methyl-N-picrylnitramine |
|---|---|
| Synonym | More Synonyms |
| Density | 1.803g/cm3 |
|---|---|
| Boiling Point | 503.7ºC at 760mmHg |
| Melting Point | 130ºC |
| Molecular Formula | C7H5N5O8 |
| Molecular Weight | 287.14300 |
| Flash Point | 258.4ºC |
| Exact Mass | 287.01400 |
| PSA | 186.52000 |
| LogP | 3.13190 |
| Index of Refraction | 1.686 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |


GHS02, GHS07 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H225-H302 + H312 + H332-H319 |
| Precautionary Statements | P210-P261-P302 + P352 + P312-P304 + P340 + P312-P337 + P313-P403 + P235 |
| Hazard Codes | E,T,F |
| Risk Phrases | 2-23/24/25-33-36/37/38-11 |
| Safety Phrases | 35-45-27-16 |
| RIDADR | 0208 |
| WGK Germany | 3 |
| Hazard Class | 1.1 |
| Precursor 10 | |
|---|---|
| DownStream 10 | |
|
Measurement of nitrosamine and nitramine formation from NOx reactions with amines during amine-based carbon dioxide capture for postcombustion carbon sequestration.
Environ. Sci. Technol. 46(17) , 9793-801, (2012) With years of full-scale experience for precombustion CO(2) capture, amine-based technologies are emerging as the prime contender for postcombustion CO(2) capture. However, concerns for postcombustion... |
|
|
Evaluation of a TiO2 photocatalysis treatment on nitrophenols and nitramines contaminated plant wastewaters by solid-phase extraction coupled with ESI HPLC-MS.
J. Hazard. Mater. 166(1) , 284-90, (2009) Nitration reactions of aromatic compounds are commonly involved in different industrial processes for pharmaceutical, pesticide or military uses. For many years, most of the manufacturing sites used l... |
|
|
A reliable simple method to estimate density of nitroaliphatics, nitrate esters and nitramines.
J. Hazard. Mater. 169(1-3) , 158-69, (2009) In this work, a new simple method is presented to estimate crystal density of three important classes of explosives including nitroalphatics, nitrate esters and nitramines. This method allows reliable... |
| N-METHYL-N,2,4,6-TETRANITROANILINE |
| Nitramine |
| 2,4,6-trinitrophenylmethylnitramine |
| EINECS 207-531-9 |
| 2,4,6-trinitrophenyl-N-methylnitramine |
| N-methyl-N-(2,4,6-trinitrophenyl)nitramide |
| 2,4,6-Tetryl |
| N-Methyl-N-picrylnitramine |
| Picrylmethylnitramine |
| Tetril |
| Tetralite |
| N-Picryl-N-methylnitramine |
| Picrylnitromethylamine |
| Tetryl |
| N-2,4,6-tetranitro-N-methylaniline |
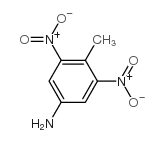 CAS#:19406-51-0
CAS#:19406-51-0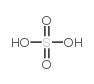 CAS#:7664-93-9
CAS#:7664-93-9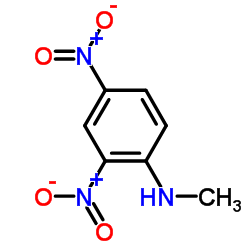 CAS#:2044-88-4
CAS#:2044-88-4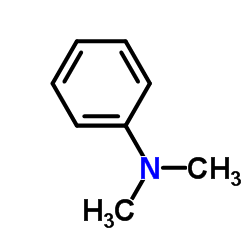 CAS#:121-69-7
CAS#:121-69-7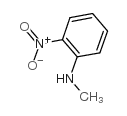 CAS#:612-28-2
CAS#:612-28-2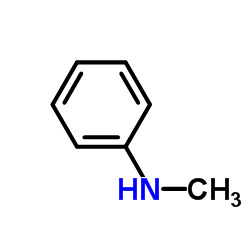 CAS#:100-61-8
CAS#:100-61-8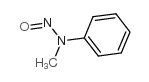 CAS#:614-00-6
CAS#:614-00-6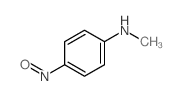 CAS#:10595-51-4
CAS#:10595-51-4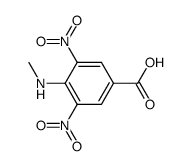 CAS#:64732-83-8
CAS#:64732-83-8 CAS#:102877-65-6
CAS#:102877-65-6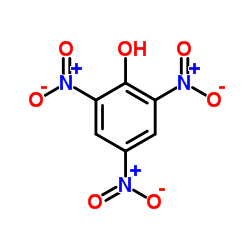 CAS#:88-89-1
CAS#:88-89-1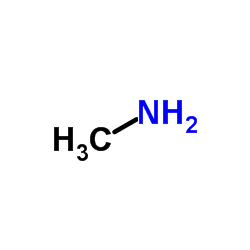 CAS#:74-89-5
CAS#:74-89-5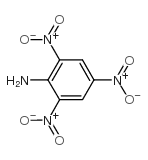 CAS#:489-98-5
CAS#:489-98-5 CAS#:7697-37-2
CAS#:7697-37-2 CAS#:7782-77-6
CAS#:7782-77-6 CAS#:1022-07-7
CAS#:1022-07-7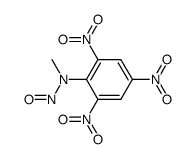 CAS#:857592-15-5
CAS#:857592-15-5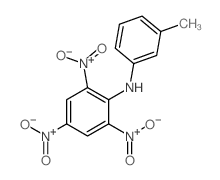 CAS#:16552-38-8
CAS#:16552-38-8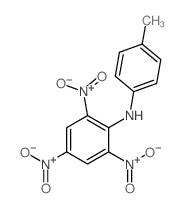 CAS#:16552-37-7
CAS#:16552-37-7 CAS#:57205-99-9
CAS#:57205-99-9