Gentisic acid
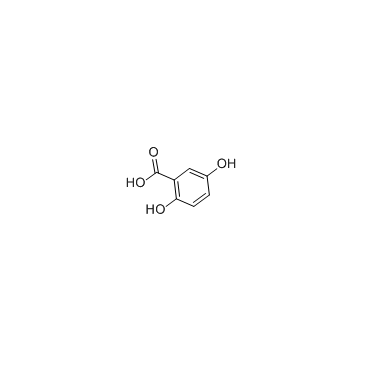
Gentisic acid structure
|
Common Name | Gentisic acid | ||
|---|---|---|---|---|
| CAS Number | 490-79-9 | Molecular Weight | 154.120 | |
| Density | 1.6±0.1 g/cm3 | Boiling Point | 406.9±35.0 °C at 760 mmHg | |
| Molecular Formula | C7H6O4 | Melting Point | 204-208 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 214.0±22.4 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of Gentisic acid2,5-Dihydroxybenzoic acid is a derivative of benzoic and a powerful inhibitor of fibroblast growth factors. |
| Name | 2,5-dihydroxybenzoic acid |
|---|---|
| Synonym | More Synonyms |
| Description | 2,5-Dihydroxybenzoic acid is a derivative of benzoic and a powerful inhibitor of fibroblast growth factors. |
|---|---|
| Related Catalog | |
| Target |
Human Endogenous Metabolite Fibroblast growth factor |
| In Vitro | 2,5-Dihydroxybenzoic acid (Gentisic acid) is a derivative of benzoic and a minor product of the metabolic break down of aspirin[1]. 2,5-Dihydroxybenzoic acid is also a component of many traditional liquors and herbal remedies, is singled out as a powerful inhibitor of fibroblast growth factors. 2,5-Dihydroxybenzoic acid is used as a lead to identify additional compounds with better inhibitory characteristics generating a new chemical class of fibroblast growth factor inhibitors that includes the agent responsible for alkaptonuria. Through low and high resolution approaches, using representative members of the fibroblast growth factor family and their cell receptors, it is shown that this class of inhibitors may employ two different mechanisms to interfere with the assembly of the signaling complexes that trigger fibroblast growth factor-driven mitogenesis[2]. |
| In Vivo | It is verified from in vivo disease models that this group of inhibitors (e.g., 2,5-Dihydroxybenzoic acid) may be of interest to treat cancer and angiogenesis-dependent diseases[2]. |
| References |
[1]. Levy G, et al. Salicylate accumulation kinetics in man. N Engl J Med. 1972 Aug 31;287(9):430-2. |
| Density | 1.6±0.1 g/cm3 |
|---|---|
| Boiling Point | 406.9±35.0 °C at 760 mmHg |
| Melting Point | 204-208 °C(lit.) |
| Molecular Formula | C7H6O4 |
| Molecular Weight | 154.120 |
| Flash Point | 214.0±22.4 °C |
| Exact Mass | 154.026611 |
| PSA | 77.76000 |
| LogP | 1.56 |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.671 |
| InChIKey | WXTMDXOMEHJXQO-UHFFFAOYSA-N |
| SMILES | O=C(O)c1cc(O)ccc1O |
| Water Solubility | soluble |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi:Irritant; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26-S36-S37/39 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | LY3850000 |
| HS Code | 29182990 |
| Precursor 10 | |
|---|---|
| DownStream 10 | |
| HS Code | 2918290000 |
|---|---|
| Summary | HS: 2918290000 other carboxylic acids with phenol function but without other oxygen function, their anhydrides, halides, peroxides, peroxyacids and their derivatives Tax rebate rate:9.0% Supervision conditions:AB(certificate of inspection for goods inward,certificate of inspection for goods outward) VAT:17.0% MFN tariff:6.5% General tariff:30.0% |
|
Structure and boosting activity of a starch-degrading lytic polysaccharide monooxygenase.
Nat. Commun. 6 , 5961, (2015) Lytic polysaccharide monooxygenases (LPMOs) are recently discovered enzymes that oxidatively deconstruct polysaccharides. LPMOs are fundamental in the effective utilization of these substrates by bact... |
|
|
Antimicrobial activity of natural products from the flora of Northern Ontario, Canada.
Pharm. Biol. 53(6) , 800-6, (2015) The number of multidrug resistant (MDR) microorganisms is increasing and the antimicrobial resistance expressed by these pathogens is generating a rising global health crisis. In fact, there are only ... |
|
|
Comparison of Mannose, Ethylene Glycol, and Methoxy-Terminated Diluents on Specificity and Selectivity of Electrochemical Peptide-Based Sensors.
Anal. Chem. 87 , 6966-73, (2015) We report the synthesis and application of three new antifouling diluents for the fabrication of an E-PB HIV sensor. Among the three thiolated antifouling diluents used in this study, the methoxy-term... |
|
Name: Binding affinity towards human GABRA1 in an in vitro assay with cellular components m...
Source: ChEMBL
Target: Gamma-aminobutyric acid receptor subunit gamma-2
External Id: CHEMBL5291799
|
|
Name: Increase in membrane potential in mollusc neurons assessed as conductance of potassiu...
Source: ChEMBL
Target: Neuron
External Id: CHEMBL3283149
|
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Binding affinity towards human ESR1 in an in vitro cell free assay (CRO assay) measur...
Source: ChEMBL
Target: Estrogen receptor
External Id: CHEMBL5291792
|
|
Name: Compound was evaluated for inhibition of human F2 in an in vitro cell free assay meas...
Source: ChEMBL
Target: Prothrombin
External Id: CHEMBL5291794
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
|
Name: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfect...
Source: Broad Institute
Target: N/A
External Id: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfected HEK293 cells Inhibition - 7011-01_Antagonist_SinglePoint_HTS_Activity
|
|
Name: Octanol-water partition coefficient, log P of the compound
Source: ChEMBL
Target: N/A
External Id: CHEMBL3283138
|
| Benzoic acid,2,5-dihydroxy |
| Gentistic acid |
| Gensigon |
| 2,5-dihydroxyphenylcarboxylic acid |
| 2,5-DHBA,DHB,Gentisic acid |
| Gensigen |
| 2,5-Dihydroxybenzoic acid |
| Gentisate |
| 2,5-Dhba |
| Hydroquinonecarboxylic acid |
| 2,5-dihydroxybenzoate |
| 5-Hydroxysalicylic acid |
| EINECS 207-718-5 |
| 2,5-Dioxybenzoic acid |
| 5-hydroxy-Salicylic acid |
| MFCD00002460 |
| 2,5-Dihydroxy-benzoic acid |
| gentisic acid |
| 3,6-Dihydroxybenzoic Acid |
| 2,5-dihydroxy benzoic acid |
| Carboxyhydroquinone |
| DHB |
| Mesalazine Impurity 7 |
 CAS#:50-79-3
CAS#:50-79-3 CAS#:89-55-4
CAS#:89-55-4 CAS#:123-31-9
CAS#:123-31-9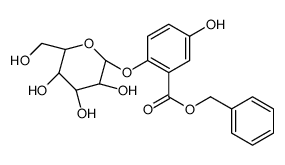 CAS#:10590-85-9
CAS#:10590-85-9 CAS#:69-72-7
CAS#:69-72-7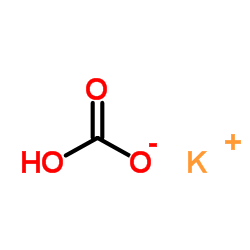 CAS#:298-14-6
CAS#:298-14-6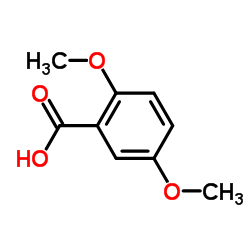 CAS#:2785-98-0
CAS#:2785-98-0 CAS#:2612-02-4
CAS#:2612-02-4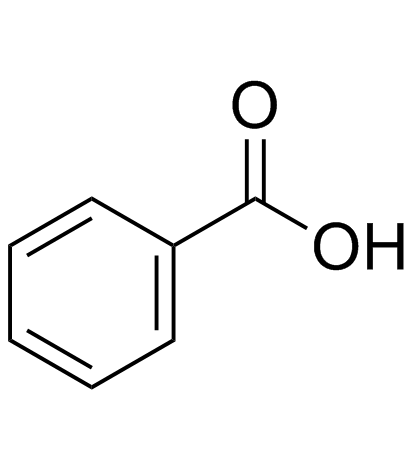 CAS#:65-85-0
CAS#:65-85-0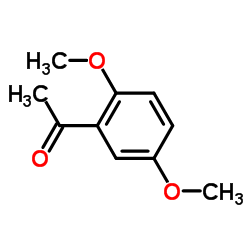 CAS#:1201-38-3
CAS#:1201-38-3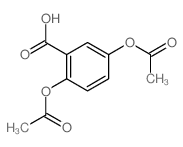 CAS#:1084-96-4
CAS#:1084-96-4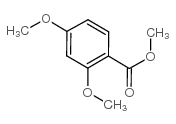 CAS#:2150-41-6
CAS#:2150-41-6 CAS#:606-45-1
CAS#:606-45-1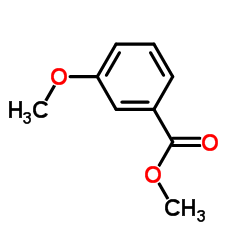 CAS#:5368-81-0
CAS#:5368-81-0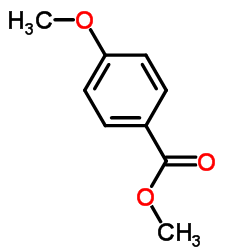 CAS#:121-98-2
CAS#:121-98-2 CAS#:2150-38-1
CAS#:2150-38-1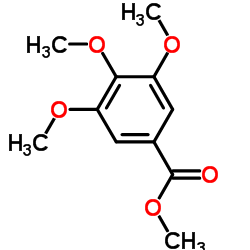 CAS#:1916-07-0
CAS#:1916-07-0 CAS#:2150-42-7
CAS#:2150-42-7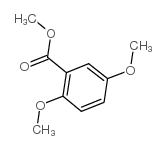 CAS#:2150-40-5
CAS#:2150-40-5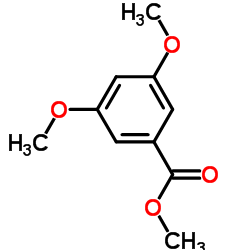 CAS#:2150-37-0
CAS#:2150-37-0
