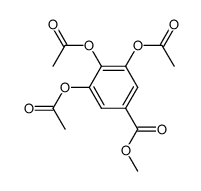
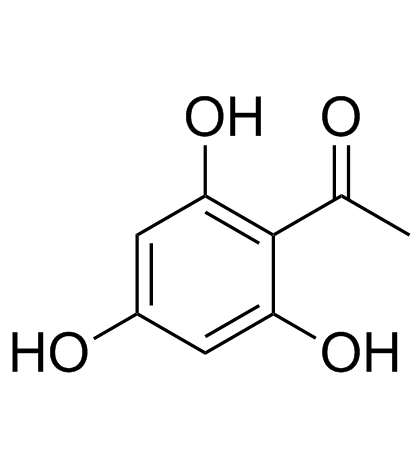
Tricetin
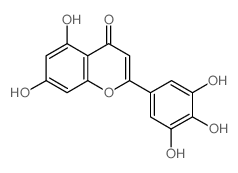
Tricetin structure
|
Common Name | Tricetin | ||
|---|---|---|---|---|
| CAS Number | 520-31-0 | Molecular Weight | 302.23600 | |
| Density | 1.763g/cm3 | Boiling Point | 721.7ºC at 760 mmHg | |
| Molecular Formula | C15H10O7 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 277.9ºC | |
Use of TricetinTricetin is a potent competitive inhibitor of the Keap1-Nrf2 Protein Protein Interaction (PPI). Tricetin protects against 6-OHDA-induced neurotoxicity in Parkinson's disease model by activating Nrf2/HO-1 signaling pathway and preventing mitochondria-dependent apoptosis pathway[1]. |
| Name | tricetin |
|---|---|
| Synonym | More Synonyms |
| Description | Tricetin is a potent competitive inhibitor of the Keap1-Nrf2 Protein Protein Interaction (PPI). Tricetin protects against 6-OHDA-induced neurotoxicity in Parkinson's disease model by activating Nrf2/HO-1 signaling pathway and preventing mitochondria-dependent apoptosis pathway[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Tricetin is mainly found in natural plants such as Ginkgo biloba L., Carica papaya L. and Murraya exotica L. Tricetin activates the Nrf2/HO-1 pathway to protect cells from oxidative stress. Tricetin possessed the protective effect on dopamine neurons of C. elegans. Tricetin has cytostatic properties and anti-metastatic activity of various solid tumors[1]. Pretreatment with Tricetin (20, 40, and 80 μM; for 4 hours) significantly improves 6-OHDA (200 μM)-induced SH-SY5Y cells viability and suppresses mitochondria-mediated apoptosis[1]. Tricetin (80 μM; for 1, 2 and 4 h) markedly decreased the expressions of p-JNK and p-p38[1]. Cell Viability Assay Cell Line: SH-SY5Y cells Concentration: 20, 40, and 80 μM Incubation Time: Pretreatment for 4 h followed by 6-OHDA (200 μM) for 24 h Result: Significantly increased 6-OHDA-induced SH-SY5Y cells viability. Western Blot Analysis Cell Line: SH-SY5Y cells Concentration: 80 μM Incubation Time: 1, 2 and 4 h Result: Markedly decreased the expressions of p-JNK and p-p38. |
| References |
| Density | 1.763g/cm3 |
|---|---|
| Boiling Point | 721.7ºC at 760 mmHg |
| Molecular Formula | C15H10O7 |
| Molecular Weight | 302.23600 |
| Flash Point | 277.9ºC |
| Exact Mass | 302.04300 |
| PSA | 131.36000 |
| LogP | 1.98800 |
| Index of Refraction | 1.804 |
| InChIKey | ARSRJFRKVXALTF-UHFFFAOYSA-N |
| SMILES | O=c1cc(-c2cc(O)c(O)c(O)c2)oc2cc(O)cc(O)c12 |
| HS Code | 2914501900 |
|---|
|
~36% 
Tricetin CAS#:520-31-0 |
| Literature: Gao, Hong; Nishida, Jun; Saito, Shizuka; Kawabata, Jun Molecules, 2007 , vol. 12, # 1 p. 86 - 97 |
|
~% 
Tricetin CAS#:520-31-0 |
| Literature: Pandurangan Letters in Organic Chemistry, 2014 , vol. 11, # 3 p. 225 - 229 |
|
~% 
Tricetin CAS#:520-31-0 |
| Literature: Anderson Canadian Journal of Research, 1932 , vol. 7, p. 285,289 Full Text Show Details Badhwar; Kang; Venkataraman Journal of the Chemical Society, 1932 , p. 1107,1108 |
|
~% 
Tricetin CAS#:520-31-0 |
| Literature: Anderson Canadian Journal of Research, 1932 , vol. 7, p. 285,289 Full Text Show Details Badhwar; Kang; Venkataraman Journal of the Chemical Society, 1932 , p. 1107,1108 |
|
~% 
Tricetin CAS#:520-31-0 |
| Literature: Bargellini; Monti Gazzetta Chimica Italiana, 1915 , vol. 45 I, p. 68 |
|
~% 
Tricetin CAS#:520-31-0 |
| Literature: Bargellini; Monti Gazzetta Chimica Italiana, 1915 , vol. 45 I, p. 68 |
| HS Code | 2914501900 |
|---|---|
| Summary | 2914501900 other ketone-phenols。Supervision conditions:None。VAT:17.0%。Tax rebate rate:9.0%。MFN tariff:5.5%。General tariff:30.0% |
|
Name: ERK5 transcriptional activity HTS
Source: 24565
Target: N/A
External Id: ERK5 transcriptional activity-HTS
|
|
Name: Cytotoxicity against human Huh7.5.1 cells by MTT assay
Source: ChEMBL
Target: N/A
External Id: CHEMBL2026691
|
|
Name: Experimentally measured binding affinity data (IC50) for protein-ligand complexes der...
Source: Shanghai Institute of Organic Chemistry
Target: N/A
External Id: PDBbind-IC50 for protein-ligand complexes
|
|
Name: Antiviral activity against HCV JFH-1 J399EM infected in Human Huh7.5.1 cells assessed...
Source: ChEMBL
Target: Hepacivirus hominis
External Id: CHEMBL2026689
|
|
Name: qHTS for Inhibitors of human tyrosyl-DNA phosphodiesterase 1 (TDP1): qHTS in cells in...
Source: NCGC
Target: TDP1 protein [Homo sapiens]
External Id: TDP1100
|
|
Name: qHTS for Stage-Specific Inhibitors of Vaccinia Orthopoxvirus: mCherry Reporter Primar...
Source: NCGC
Target: 67.9K protein [Vaccinia virus]
External Id: Vaccinia-p2mCherry
|
|
Name: qHTS for Inhibitors of human tyrosyl-DNA phosphodiesterase 1 (TDP1): qHTS in cells in...
Source: NCGC
Target: TDP1 protein [Homo sapiens]
External Id: TDP1101
|
|
Name: Spectrum HTS for Inhibitors of Aerobactin Synthetase IucA
Source: 23265
Target: IucA Synthetase from hypervirulent Klebsiella pneumoniae hvKP1
External Id: IucA Pilot Assay Spectrum Library
|
|
Name: Inhibition of sodium fluorescein uptake in OATP1B3-transfected CHO cells at an equimo...
Source: ChEMBL
Target: Solute carrier organic anion transporter family member 1B3
External Id: CHEMBL3039491
|
| 5,7,3',4',5'-Pentahydroxyflavone |
| Hieracin |
| Tricetin |
| 5,7-dihydroxy-2-(3,4,5-trihydroxyphenyl)chromen-4-one |