Harmalane
Modify Date: 2025-08-25 18:19:52
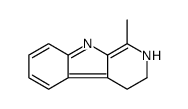
Harmalane structure
|
Common Name | Harmalane | ||
|---|---|---|---|---|
| CAS Number | 525-41-7 | Molecular Weight | 184.24 | |
| Density | 1.23g/cm3 | Boiling Point | 390.3ºC at 760mmHg | |
| Molecular Formula | C12H12N2 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 189.9ºC | |
Use of HarmalaneHarmalan (compound 8) is a alkaloid isolated from the extracts of the leaves of Flindersia laevicarpa[1]. |
| Name | 1-methyl-3,4-dihydro-2H-pyrido[3,4-b]indole |
|---|---|
| Synonym | More Synonyms |
| Description | Harmalan (compound 8) is a alkaloid isolated from the extracts of the leaves of Flindersia laevicarpa[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.23g/cm3 |
|---|---|
| Boiling Point | 390.3ºC at 760mmHg |
| Molecular Formula | C12H12N2 |
| Molecular Weight | 184.24 |
| Flash Point | 189.9ºC |
| Exact Mass | 184.10000 |
| PSA | 24.39000 |
| LogP | 0.45970 |
| Index of Refraction | 1.678 |
| Hazard Codes | Xi |
|---|---|
| HS Code | 2933990090 |
| Precursor 9 | |
|---|---|
| DownStream 8 | |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Name: Primary cell-based high-throughput screening assay for identification of compounds th...
Source: Johns Hopkins Ion Channel Center
Target: regulator of G-protein signaling 4 isoform 2 [Homo sapiens]
External Id: JHICC_RGS_Act_HTS
|
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: uHTS identification of small molecule activators of the adaptive arm of the Unfolded ...
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: BCCG-A405-UPR-XBP1-PrimaryAgonist-Assay
|
|
Name: High throughput fluorescence intensity-based biochemical assay to screen for small mo...
Source: University of Pittsburgh Molecular Library Screening Center
Target: furin (paired basic amino acid cleaving enzyme), isoform CRA_a [Homo sapiens]
External Id: MH080376 Biochemical HTS for Inhibitors of the Proprotein Convertase Furin.
|
|
Name: Fluorescence polarization to screen for inhibitor that competite the binding of FadD2...
Source: Broad Institute
Target: FATTY-ACID-CoA LIGASE FADD28 (FATTY-ACID-CoA SYNTHETASE)
External Id: 2147-01_Inhibitor_SinglePoint_HTS_Activity
|
|
Name: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfect...
Source: Broad Institute
Target: N/A
External Id: Bursicon-induced LGR2 mediated cAMP production in LGR-2/CRE6x-Luciferase co-transfected HEK293 cells Inhibition - 7011-01_Antagonist_SinglePoint_HTS_Activity
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: Confirmed inhibitors of the Choline Transporter (CHT)
Source: 1043
Target: high affinity choline transporter 1 [Homo sapiens]
External Id: SAID_488997
|
Total 130, Current Page 1 of 13
1
2
3
4
5
| Harmalan |
![1H-Pyrido[3,4-b]indole,2,3,4,9-tetrahydro-1-methyl structure](https://image.chemsrc.com/caspic/473/2506-10-7.png)
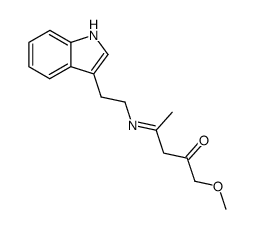
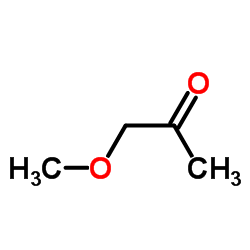
![2-[1-[[2-(1H-Indol-3-yl)ethyl]amino]ethylidene]cyclohexanone structure](https://image.chemsrc.com/caspic/127/107292-45-5.png)

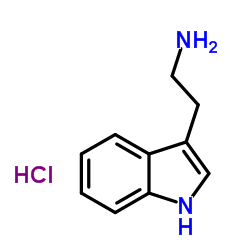
![1H-Pyrido[3,4-b]indole-1-carboxylicacid, 2,3,4,9-tetrahydro-1-methyl structure](https://image.chemsrc.com/caspic/106/6543-83-5.png)

 CAS#:479-43-6
CAS#:479-43-6 CAS#:486-84-0
CAS#:486-84-0![3-ethyl-7,12-dihydro-6H-indolo[2,3-a]quinolizin-4-one structure](https://image.chemsrc.com/caspic/110/69303-10-2.png) CAS#:69303-10-2
CAS#:69303-10-2![3-acetyl-7,12-dihydro-6H-indolo[2,3-a]quinolizin-4-one structure](https://image.chemsrc.com/caspic/228/89652-82-4.png) CAS#:89652-82-4
CAS#:89652-82-4![1-(1-methylidene-4,9-dihydro-3H-pyrido[3,4-b]indol-2-yl)ethanone structure](https://image.chemsrc.com/caspic/484/61828-58-8.png) CAS#:61828-58-8
CAS#:61828-58-8![methyl 6-oxo-6h-indolo[3,2,1-de][1,5]naphthyridine-5-carboxylate structure](https://image.chemsrc.com/caspic/428/55854-61-0.png) CAS#:55854-61-0
CAS#:55854-61-0 CAS#:525-40-6
CAS#:525-40-6![(S)-1-methyl-1,2,3,4-tetrahydro-9H-pyrido[3,4-b]indole structure](https://image.chemsrc.com/caspic/233/23844-21-5.png) CAS#:23844-21-5
CAS#:23844-21-5