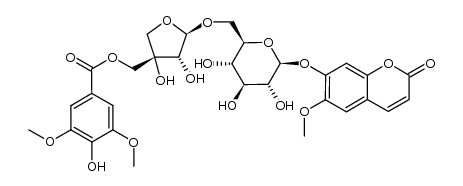
scopolin
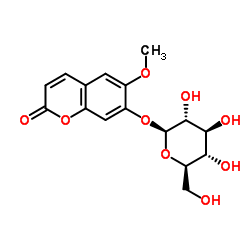
scopolin structure
|
Common Name | scopolin | ||
|---|---|---|---|---|
| CAS Number | 531-44-2 | Molecular Weight | 354.309 | |
| Density | 1.6±0.1 g/cm3 | Boiling Point | 650.2±55.0 °C at 760 mmHg | |
| Molecular Formula | C16H18O9 | Melting Point | 217ºC | |
| MSDS | N/A | Flash Point | 241.5±25.0 °C | |
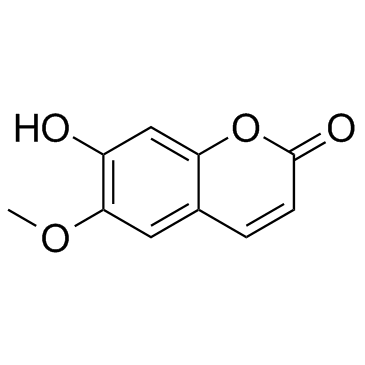
Use of scopolinScopolin is a coumarin isolated from Arabidopsis thaliana (Arabidopsis) roots[1]. Scopolin attenuated hepatic steatosis through activation of SIRT1-mediated signaling cascades[2]. |
| Name | scopolin |
|---|---|
| Synonym | More Synonyms |
| Description | Scopolin is a coumarin isolated from Arabidopsis thaliana (Arabidopsis) roots[1]. Scopolin attenuated hepatic steatosis through activation of SIRT1-mediated signaling cascades[2]. |
|---|---|
| Related Catalog | |
| Target |
SIRT1 |
| References |
| Density | 1.6±0.1 g/cm3 |
|---|---|
| Boiling Point | 650.2±55.0 °C at 760 mmHg |
| Melting Point | 217ºC |
| Molecular Formula | C16H18O9 |
| Molecular Weight | 354.309 |
| Flash Point | 241.5±25.0 °C |
| Exact Mass | 354.095093 |
| PSA | 138.82000 |
| LogP | -1.87 |
| Vapour Pressure | 0.0±2.0 mmHg at 25°C |
| Index of Refraction | 1.639 |
| InChIKey | SGTCGCCQZOUMJJ-YMILTQATSA-N |
| SMILES | COc1cc2ccc(=O)oc2cc1OC1OC(CO)C(O)C(O)C1O |
|
~% 
scopolin CAS#:531-44-2 |
| Literature: Chemistry of Natural Compounds, , vol. 18, # 6 p. 654 - 657 Khimiya Prirodnykh Soedinenii, , vol. 18, # 6 p. 691 - 695 |
|
~% 
scopolin CAS#:531-44-2 |
| Literature: Chemistry of Natural Compounds, , vol. 16, # 2 p. 125 - 128 Khimiya Prirodnykh Soedinenii, , vol. 16, # 2 p. 168 - 172 |
|
~% 
scopolin CAS#:531-44-2 |
| Literature: Angewandte Chemie - International Edition, , vol. 45, # 21 p. 3534 - 3538 |
|
~% 
scopolin CAS#:531-44-2 |
| Literature: Song, Shuang; Li, Yixiu; Feng, Ziming; Jiang, Jianshuang; Zhang, Peicheng Journal of Natural Products, 2010 , vol. 73, # 2 p. 177 - 184 |
| Scopolin |
| Scopoletin glucoside |
| 7-(β-D-glucopyranosoyloxy)-6-methoxy-2H-1-benzopyran-2-one |
| 5Mg |
| 7-(b-D-Glucopyranosyloxy)-6-methoxy-2H-1-benzopyran-2-one |
| Scopoletin 7-O-Glucoside |
| 6-Methoxy-2-oxo-2H-chromen-7-yl β-D-glucopyranoside |
| 2'-β-D-glucosyl scopolin |
| Murrayin |
| Scopolin (8CI) |
| CPD-678 |
| Scopoletin 7-glucoside |
| 7β-D-Glucopyranosyloxy-6-methoxycoumarin |
| Scopoline |
| Scopoloside |

 CAS#:492-61-5
CAS#:492-61-5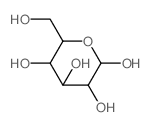 CAS#:2280-44-6
CAS#:2280-44-6
