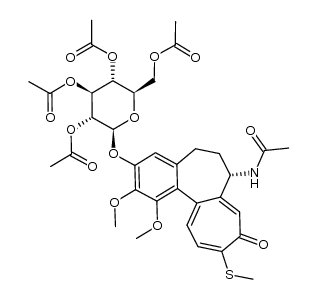
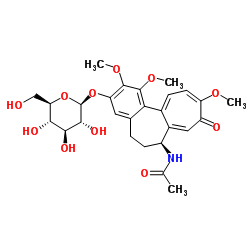
Thiocolchicoside

Thiocolchicoside structure
|
Common Name | Thiocolchicoside | ||
|---|---|---|---|---|
| CAS Number | 602-41-5 | Molecular Weight | 563.617 | |
| Density | 1.5±0.1 g/cm3 | Boiling Point | 929.6±65.0 °C at 760 mmHg | |
| Molecular Formula | C27H33NO10S | Melting Point | 190-198ºC | |
| MSDS | Chinese USA | Flash Point | 516.0±34.3 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of ThiocolchicosideThiocolchicoside is a competitive γ-aminobutyric acid type A (GABAA) receptor antagonist and glycine receptor agonist in the central nervous system. Thiocolchicoside is a semisynthetic sulfur derivative of colchicoside. Thiocolchicoside is a muscle relaxant and has anti-inflammatory, and analgesic properties[1]. |
| Name | Thiocolchicoside |
|---|---|
| Synonym | More Synonyms |
| Description | Thiocolchicoside is a competitive γ-aminobutyric acid type A (GABAA) receptor antagonist and glycine receptor agonist in the central nervous system. Thiocolchicoside is a semisynthetic sulfur derivative of colchicoside. Thiocolchicoside is a muscle relaxant and has anti-inflammatory, and analgesic properties[1]. |
|---|---|
| Related Catalog | |
| Target |
GABAA) receptor; glycine receptor[1] |
| In Vitro | Thiocolchicoside (0.001, 0.1, 1, 10, 100 μM) reduces the amplitude of eIPSCs in a concentration-dependent manner with this effect being significant at 0.1 μM and maximal at 10 μM[1]. |
| References |
| Density | 1.5±0.1 g/cm3 |
|---|---|
| Boiling Point | 929.6±65.0 °C at 760 mmHg |
| Melting Point | 190-198ºC |
| Molecular Formula | C27H33NO10S |
| Molecular Weight | 563.617 |
| Flash Point | 516.0±34.3 °C |
| Exact Mass | 563.182495 |
| PSA | 189.31000 |
| LogP | -1.23 |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.657 |
| InChIKey | LEQAKWQJCITZNK-AXHKHJLKSA-N |
| SMILES | COc1c(OC2OC(CO)C(O)C(O)C2O)cc2c(c1OC)-c1ccc(SC)c(=O)cc1C(NC(C)=O)CC2 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H315-H319-H335 |
| Precautionary Statements | P301 + P312 + P330-P305 + P351 + P338 |
| RIDADR | NONH for all modes of transport |
| RTECS | AC2976015 |
|
~96% 
Thiocolchicoside CAS#:602-41-5 |
| Literature: Gelmi, Maria Luisa; Fontana, Gabriele; Pocar, Donato; Pontremoli, Guido; Pellegrino, Sara; Bombardelli, Ezio; Riva, Antonella; Balduini, Walter; Carloni, Silvia; Cimino, Mauro Journal of Medicinal Chemistry, 2007 , vol. 50, # 9 p. 2245 - 2248 |
|
~% 
Thiocolchicoside CAS#:602-41-5 |
| Literature: Journal of Medicinal Chemistry, , vol. 50, # 9 p. 2245 - 2248 |
|
~% 
Thiocolchicoside CAS#:602-41-5 |
| Literature: Journal of Medicinal Chemistry, , vol. 50, # 9 p. 2245 - 2248 |
|
~% 
Thiocolchicoside CAS#:602-41-5 |
| Literature: Bulletin de la Societe Chimique de France, , p. 198 |
|
~% 
Thiocolchicoside CAS#:602-41-5 |
| Literature: Bulletin de la Societe Chimique de France, , p. 198 |
|
Name: Compound was evaluated for inhibition of rat Gabra1 in an in vitro assay with cellula...
Source: ChEMBL
Target: Gamma-aminobutyric acid receptor subunit alpha-1
External Id: CHEMBL5291801
|
|
Name: Binding affinity towards rat Gabra1 in an in vitro assay with cellular components mea...
Source: ChEMBL
Target: Gamma-aminobutyric acid receptor subunit alpha-1
External Id: CHEMBL5291798
|
|
Name: Binding affinity towards human ESR1 in an in vitro cell free assay (NIBR assay) measu...
Source: ChEMBL
Target: Estrogen receptor
External Id: CHEMBL5291791
|
|
Name: Binding affinity towards human EDNRA in an in vitro assay with cellular components me...
Source: ChEMBL
Target: Endothelin-1 receptor
External Id: CHEMBL5291785
|
|
Name: Dicer-mediated maturation of pre-microRNA
Source: Center for Chemical Genomics, University of Michigan
Target: N/A
External Id: TargetID_659_CEMA
|
|
Name: Binding affinity towards human DRD2 in an in vitro assay with cellular components mea...
Source: ChEMBL
Target: D(2) dopamine receptor
External Id: CHEMBL5291781
|
|
Name: Agonist activity at human DRD1 in an in vitro cell-based assay measured by time-resol...
Source: ChEMBL
Target: D(1A) dopamine receptor
External Id: CHEMBL5291777
|
|
Name: Antagonist activity at human CNR1 in an in vitro cell-based assay measured by fluores...
Source: ChEMBL
Target: Cannabinoid receptor 1
External Id: CHEMBL5291772
|
|
Name: Agonist activity at human CNR1 in an in vitro cell-based assay measured by fluorescen...
Source: ChEMBL
Target: Cannabinoid receptor 1
External Id: CHEMBL5291771
|
|
Name: qHTS assay to test for compound auto fluorescence at 535 nm (green) in HEK293 cell fr...
Source: 824
Target: N/A
External Id: SPEC167MG
|
| Thiocolchicoside |
| UNII:T1X8S697GT |
| Thiocolchicine Glycoside |
| N-[(7S)-3-(b-D-glucopyranosyloxy)-1,2-dimethoxy-10-(methylthio)-9-oxo-5,6,7,9-tetrahydrobenzo[a]heptalen-7-yl]acetamide |
| R. 271 |
| Coltramyl |
| Musco-ril |
| N-[(7S)-3-(β-D-Glucopyranosyloxy)-1,2-dimethoxy-10-(methylsulfanyl)-9-oxo-5,6,7,9-tetrahydrobenzo[a]heptalen-7-yl]acetamide |
| 3,10-Di(demethoxy)-3-glucosyloxy-10-methylthiocolchicine |
| 10-Thiocolchicoside |
| Tiocolchicoside |