Gliotoxin
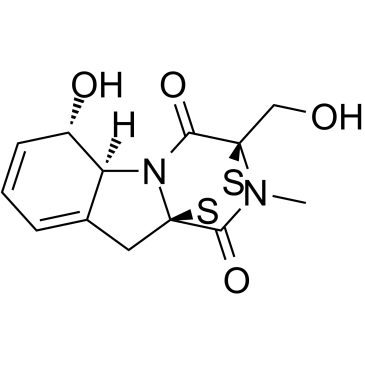
Gliotoxin structure
|
Common Name | Gliotoxin | ||
|---|---|---|---|---|
| CAS Number | 67-99-2 | Molecular Weight | 326.391 | |
| Density | 1.8±0.1 g/cm3 | Boiling Point | 699.7±55.0 °C at 760 mmHg | |
| Molecular Formula | C13H14N2O4S2 | Melting Point | 153.5ºC | |
| MSDS | Chinese USA | Flash Point | 377.0±31.5 °C | |
| Symbol |


GHS02, GHS07 |
Signal Word | Danger | |
Use of GliotoxinGliotoxin is a secondary metabolite, the most abundant mycotoxin secreted by A. fumigatus, inhibits the phagocytosis of macrophages and the immune functions of other immune cells [1]. Gliotoxin inhibits inducible NF-κB activity by preventing IκB degradation, which consequently induces host-cell apoptosis[2]. Gliotoxin activates PKA and increases intracellular cAMP concentration; modulates actin cytoskeleton rearrangement to facilitate A. fumigatus internalization into lung epithelial cells[3]. |
| Name | gliotoxin |
|---|---|
| Synonym | More Synonyms |
| Description | Gliotoxin is a secondary metabolite, the most abundant mycotoxin secreted by A. fumigatus, inhibits the phagocytosis of macrophages and the immune functions of other immune cells [1]. Gliotoxin inhibits inducible NF-κB activity by preventing IκB degradation, which consequently induces host-cell apoptosis[2]. Gliotoxin activates PKA and increases intracellular cAMP concentration; modulates actin cytoskeleton rearrangement to facilitate A. fumigatus internalization into lung epithelial cells[3]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.8±0.1 g/cm3 |
|---|---|
| Boiling Point | 699.7±55.0 °C at 760 mmHg |
| Melting Point | 153.5ºC |
| Molecular Formula | C13H14N2O4S2 |
| Molecular Weight | 326.391 |
| Flash Point | 377.0±31.5 °C |
| Exact Mass | 326.039490 |
| PSA | 131.68000 |
| LogP | 0.52 |
| Vapour Pressure | 0.0±5.0 mmHg at 25°C |
| Index of Refraction | 1.814 |
| InChIKey | FIVPIPIDMRVLAY-RBJBARPLSA-N |
| SMILES | CN1C(=O)C23CC4=CC=CC(O)C4N2C(=O)C1(CO)SS3 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |


GHS02, GHS07 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H225-H302-H312-H319-H332 |
| Precautionary Statements | P210-P280-P305 + P351 + P338 |
| Personal Protective Equipment | Eyeshields;Faceshields;Gloves;type P2 (EN 143) respirator cartridges |
| Hazard Codes | T |
| Risk Phrases | 25 |
| Safety Phrases | S45;S36/S37/S39 |
| RIDADR | UN 3462 6.1/PG 3 |
| WGK Germany | 3 |
| RTECS | KB4725000 |
| Packaging Group | III |
| Hazard Class | 6.1(b) |
| HS Code | 29419090 |
|
The MAP kinase MpkA controls cell wall integrity, oxidative stress response, gliotoxin production and iron adaptation in Aspergillus fumigatus.
Mol. Microbiol. 82(1) , 39-53, (2011) The saprophytic fungus Aspergillus fumigatus is the most important air-borne fungal pathogen. The cell wall of A. fumigatus has been studied intensively as a potential target for development of effect... |
|
|
Single-pot derivatisation strategy for enhanced gliotoxin detection by HPLC and MALDI-ToF mass spectrometry.
Anal. Bioanal. Chem 401(8) , 2519-29, (2011) Gliotoxin is produced by non-ribosomal peptide synthesis and secreted from certain fungi, including Aspergillus fumigatus. It is an epipolythiodioxopiperazine that contains an intact disulphide bridge... |
|
|
Individual and combined effects of mycotoxins from typical indoor moulds.
Toxicol. In Vitro 27(6) , 1970-8, (2013) The mycotoxins patulin, gliotoxin and sterigmatocystin can be produced by common indoor moulds and enter the human body via inhalation of mycotoxin containing spores and particulates. There are variou... |
|
Name: Antimicrobial activity against Saccharomyces cerevisiae containing disruption in YKL0...
Source: ChEMBL
Target: Saccharomyces cerevisiae
External Id: CHEMBL1262461
|
|
Name: Antimicrobial activity against Saccharomyces cerevisiae containing disruption in MEF2...
Source: ChEMBL
Target: Saccharomyces cerevisiae
External Id: CHEMBL1262460
|
|
Name: Antimicrobial activity against Saccharomyces cerevisiae containing disruption in CYS3...
Source: ChEMBL
Target: Saccharomyces cerevisiae
External Id: CHEMBL1262459
|
|
Name: Antimicrobial activity against Saccharomyces cerevisiae containing disruption in YGL0...
Source: ChEMBL
Target: Saccharomyces cerevisiae
External Id: CHEMBL1262458
|
|
Name: USP8 deubiquitinase inhibition: Primary qHTS
Source: 24642
Target: ubiquitin specific peptidase 8
External Id: USP8 FAST DUB HTS Primary
|
|
Name: Antimicrobial activity against Saccharomyces cerevisiae containing disruption in YLR4...
Source: ChEMBL
Target: Saccharomyces cerevisiae
External Id: CHEMBL1262457
|
|
Name: Antimicrobial activity against Saccharomyces cerevisiae containing disruption in YOR3...
Source: ChEMBL
Target: Saccharomyces cerevisiae
External Id: CHEMBL1262456
|
|
Name: USP17 deubiquitinase inhibition: Primary qHTS
Source: 24642
Target: ubiquitin specific peptidase 17 like family member 5
External Id: USP17 FAST DUB HTS Primary
|
|
Name: Antimicrobial activity against Saccharomyces cerevisiae containing disruption in YGL2...
Source: ChEMBL
Target: Saccharomyces cerevisiae
External Id: CHEMBL1262455
|
|
Name: Antimicrobial activity against fluconazole-resistant Cryptococcus gattii serotype C i...
Source: ChEMBL
Target: Cryptococcus gattii
External Id: CHEMBL1261944
|
| (1R,7S,8S,11R)-7-Hydroxy-11-(hydroxymethyl)-15-methyl-12,13-dithia-9,15-diazatetracyclo[9.2.2.0.0]pentadeca-3,5-diene-10,14-dione |
| epithiodiketopiperazine |
| Gliotoxin,(3R,5aS,6S,10aR)-2,3,5a,6-Tetrahydro-6-hydroxy-3-(hydroxymethyl)-2-methyl-10H-3,10a-epidithiopyrazino[1,2-a]indole-1,4-dione |
| MFCD00058534 |
| (3R,5aS,6S,10aR)-6-hydroxy-3-(hydroxymethyl)-2-methyl-2,3,6,10-tetrahydro-5aH-3,10a-epidithiopyrazino[1,2-a]indole-1,4-dione |
| [3R-(3a,5ab,6b,10aa)]-2,3,5a,6-Tetrahydro-6-hydroxy-3-(hydroxymethyl)-2-methyl-10H3,10a-epidithiopyrazino[1,2-a]indole-1,4-dione |
| Gliotoxin |
![(3aS,5S,6aR,7S,9aR)-6-tert-butyl 5-methyl 2-thioxo-7-((triisopropylsilyl)oxy)-4,5,6a,7-tetrahydro-[1,3]dioxolo[4,5-d]indole-5,6(9aH)-dicarboxylate Structure](https://image.chemsrc.com/caspic/135/1402592-69-1.png) CAS#:1402592-69-1
CAS#:1402592-69-1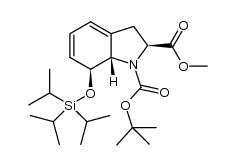 CAS#:1402592-87-3
CAS#:1402592-87-3![Pyrazino[1,2-a]indole-1,4-dione,2,3-dihydro-2-methyl-3-methylene structure](https://image.chemsrc.com/caspic/271/19079-11-9.png) CAS#:19079-11-9
CAS#:19079-11-9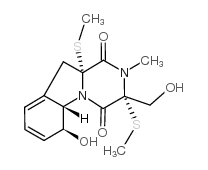 CAS#:74149-38-5
CAS#:74149-38-5
