Bepridil hydrochloride
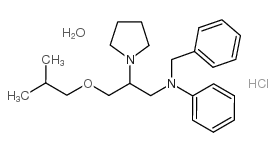
Bepridil hydrochloride structure
|
Common Name | Bepridil hydrochloride | ||
|---|---|---|---|---|
| CAS Number | 74764-40-2 | Molecular Weight | 421.01600 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C24H37ClN2O2 | Melting Point | 91 ±2° | |
| MSDS | USA | Flash Point | N/A | |
Use of Bepridil hydrochlorideBepridil hydrochloride hydrate ((±)-Bepridil hydrochloride hydrate) is a non-selective, long-acting Ca+ channel antagonist and Na+, K+ channel inhibitor, with antianginal and type I antiarrhythmic effects. Bepridil hydrochloride hydrate also acts as a cardiac Na+/Ca2+ exchange (NCX1) inhibitor. Bepridil hydrochloride hydrate can be used for the research of cardiovascular disorders[1][2][3][4][5]. |
| Name | bepridil hydrochloride monohydrate |
|---|---|
| Synonym | More Synonyms |
| Description | Bepridil hydrochloride hydrate ((±)-Bepridil hydrochloride hydrate) is a non-selective, long-acting Ca+ channel antagonist and Na+, K+ channel inhibitor, with antianginal and type I antiarrhythmic effects. Bepridil hydrochloride hydrate also acts as a cardiac Na+/Ca2+ exchange (NCX1) inhibitor. Bepridil hydrochloride hydrate can be used for the research of cardiovascular disorders[1][2][3][4][5]. |
|---|---|
| Related Catalog | |
| Target |
Ca2+ |
| In Vitro | Bepridil hydrochloride hydrate blockades of Ca2+-dependent action potentials in vascular smooth muscle of dog coronary artery[2]. Bepridil hydrochloride hydrate blocks Ca currents and Na currents with IC50s of 0.5 μM and 30 μM, respectively in cultured ventricular cells[4]. Bepridil hydrochloride hydrate decreases IKs under blockade of IKr with E4031 (5 μM), in a concentration-dependent manner[5]. |
| In Vivo | Bepridil hydrochloride hydrate exhibits a half-life of averages 33±15 hours after single-dose administration[6]. |
| References |
[1]. A Gill, et al. Pharmacology of Bepridil. Am J Cardiol. 1992 Apr 9;69(11):11D-16D. |
| Melting Point | 91 ±2° |
|---|---|
| Molecular Formula | C24H37ClN2O2 |
| Molecular Weight | 421.01600 |
| Exact Mass | 420.25400 |
| PSA | 24.94000 |
| LogP | 5.50580 |
| Appearance of Characters | powder | white |
| InChIKey | UEECHQPWQHYEDE-UHFFFAOYSA-N |
| SMILES | CC(C)COCC(CN(Cc1ccccc1)c1ccccc1)N1CCCC1.Cl.O |
| Storage condition | Desiccate at RT |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 2 |
| RTECS | UY1162000 |
|
Pulsed infrared radiation excites cultured neonatal spiral and vestibular ganglion neurons by modulating mitochondrial calcium cycling.
J. Neurophysiol. 112(6) , 1246-55, (2014) Cochlear implants are currently the most effective solution for profound sensorineural hearing loss, and vestibular prostheses are under development to treat bilateral vestibulopathies. Electrical cur... |
|
|
A recoverable state of axon injury persists for hours after spinal cord contusion in vivo.
Nat. Commun. 5 , 5683, (2014) Therapeutic strategies for spinal cord injury (SCI) commonly focus on regenerating disconnected axons. An alternative approach would be to maintain continuity of damaged axons, especially after contus... |
|
|
Serum flecainide S/R ratio reflects the CYP2D6 genotype and changes in CYP2D6 activity.
Drug Metab. Pharmacokinet. 30 , 257-62, (2015) The aims of this study were to clarify whether the ratio of S- to R-flecainide (S/R ratio) in the serum flecainide concentration was associated with the stereoselectivity of flecainide metabolism, and... |
|
Name: Identifying Sarm1 Tir Hydrolase inhibitors through NAD-Glo assay
Source: 24386
Target: N/A
External Id: Sarm1 Tir NADase inhibitors screen
|
|
Name: Inhibition of sodium fluorescein uptake in OATP1B1-transfected CHO cells at an equimo...
Source: ChEMBL
Target: Solute carrier organic anion transporter family member 1B1
External Id: CHEMBL3039488
|
|
Name: Inhibition of neurosphere proliferation of mouse neural precursor cells by MTT assay
Source: ChEMBL
Target: N/A
External Id: CHEMBL1266185
|
|
Name: Drug Induced Liver Injury Prediction System (DILIps) validation dataset; compound DIL...
Source: ChEMBL
Target: Hepatotoxicity
External Id: CHEMBL1909323
|
|
Name: Literature-mined public compounds from Greene et al multi-species hepatotoxicity mode...
Source: ChEMBL
Target: Hepatotoxicity
External Id: CHEMBL1697722
|
|
Name: Human drug-induced liver injury (DILI) modelling dataset from Ekins et al
Source: ChEMBL
Target: Hepatotoxicity
External Id: CHEMBL1697730
|
|
Name: Inhibition of sodium fluorescein uptake in OATP1B3-transfected CHO cells at an equimo...
Source: ChEMBL
Target: Solute carrier organic anion transporter family member 1B3
External Id: CHEMBL3039491
|
| 3-isobutoxy-2-pyrrolidino-n-phenyl-n-benzylpropylaminehydrochloridehydrate |
| MFCD00065498 |
| Unii-4W2p15D93m |
| l)-,monohydrochloride,monohydrate |
| angopril |
| Bepridil hydrochloride,N-Benzyl-N-(3-isobutoxy-2-pyrrolidin-1-yl-propyl)anilinehydrochloride |