L-Prolinamide
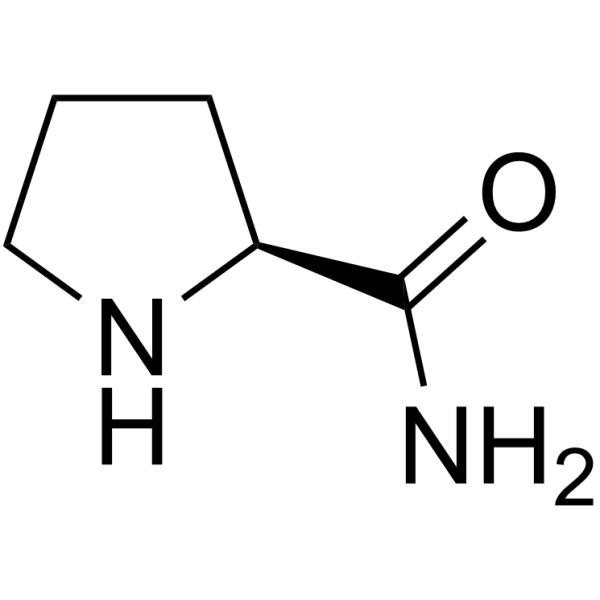
L-Prolinamide structure
|
Common Name | L-Prolinamide | ||
|---|---|---|---|---|
| CAS Number | 7531-52-4 | Molecular Weight | 114.146 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 303.6±31.0 °C at 760 mmHg | |
| Molecular Formula | C5H10N2O | Melting Point | 95-97 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 137.4±24.8 °C | |
Use of L-ProlinamideL-Prolinamide is a proline derivative[1]. |
| Name | L-prolinamide |
|---|---|
| Synonym | More Synonyms |
| Description | L-Prolinamide is a proline derivative[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Amino acids and amino acid derivatives have been commercially used as ergogenic supplements. They influence the secretion of anabolic hormones, supply of fuel during exercise, mental performance during stress related tasks and prevent exercise induced muscle damage. They are recognized to be beneficial as ergogenic dietary substances[1]. |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 303.6±31.0 °C at 760 mmHg |
| Melting Point | 95-97 °C(lit.) |
| Molecular Formula | C5H10N2O |
| Molecular Weight | 114.146 |
| Flash Point | 137.4±24.8 °C |
| Exact Mass | 114.079315 |
| PSA | 55.12000 |
| LogP | -1.51 |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.491 |
| InChIKey | VLJNHYLEOZPXFW-BYPYZUCNSA-N |
| SMILES | NC(=O)C1CCCN1 |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xn: Harmful; |
| Risk Phrases | R22 |
| Safety Phrases | S22-S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 2933990090 |
| Precursor 9 | |
|---|---|
| DownStream 10 | |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Transport and signaling via the amino acid binding site of the yeast Gap1 amino acid transceptor.
Nat. Chem. Biol. 5 , 45-52, (2009) Transporter-related nutrient sensors, called transceptors, mediate nutrient activation of signaling pathways through the plasma membrane. The mechanism of action of transporting and nontransporting tr... |
|
|
Enantioseparation of amino acids and alpha-hydroxy acids on ligand-exchange continuous beds by capillary electrochromatography.
Electrophoresis 31 , 1517-1520, (2010) A new chiral stationary phase based on continuous bed (CB) technology using L-prolinamide as a chiral selector was prepared. Its ability for enantioseparation of amino acids and alpha-hydroxy acids by... |
|
|
d-Glucosamine in a chimeric prolinamide organocatalyst for direct asymmetric aldol addition
Carbohydr. Res. 356 , 273-7, (2012) O-TBDPS D-glucosamine coupled with l-proline is reported to act as an efficient organocatalyst in the accomplishment of direct aldol reactions. Excellent results, in terms of chemical yields, as well ... |
| (S)-Prolinamide |
| (2S)-2-Pyrrolidinecarboxamide |
| h-pro-nh2 |
| (S)-Pyrrolidine-2-carboxylic acid amide |
| MFCD00005253 |
| (2S)-2-Pyrrolidinecarboximidic acid |
| EINECS 231-397-0 |
| (S)-pyrrolidine-2-carboxamide |
| L-Prolinamide |
| (2S)-pyrrolidine-2-carboxamide |
| Prolinamide |
| L-proline amide |
| L-Prolineamide |
 CAS#:34079-31-7
CAS#:34079-31-7 CAS#:35150-07-3
CAS#:35150-07-3 CAS#:2577-48-2
CAS#:2577-48-2 CAS#:147-85-3
CAS#:147-85-3 CAS#:2133-40-6
CAS#:2133-40-6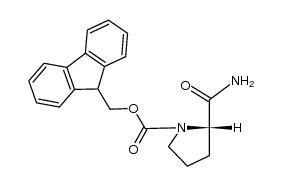 CAS#:115134-39-9
CAS#:115134-39-9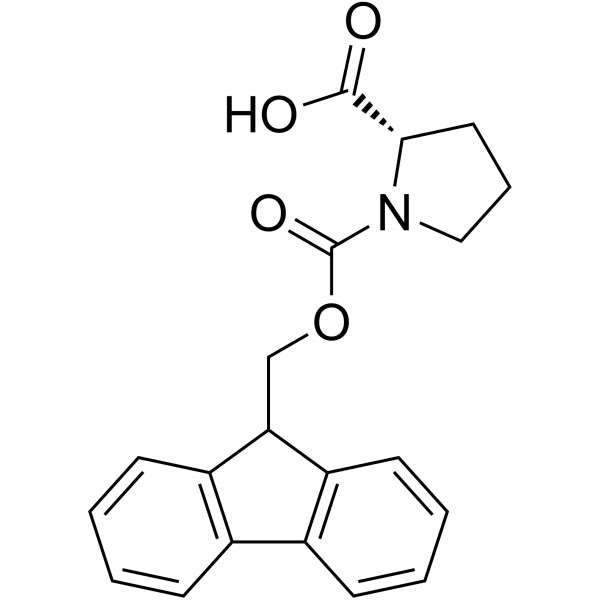 CAS#:71989-31-6
CAS#:71989-31-6 CAS#:7732-18-5
CAS#:7732-18-5 CAS#:17460-56-9
CAS#:17460-56-9 CAS#:59936-29-7
CAS#:59936-29-7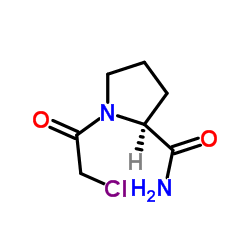 CAS#:214398-99-9
CAS#:214398-99-9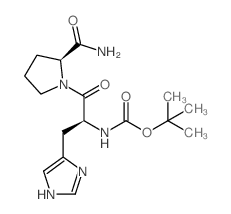 CAS#:29133-55-9
CAS#:29133-55-9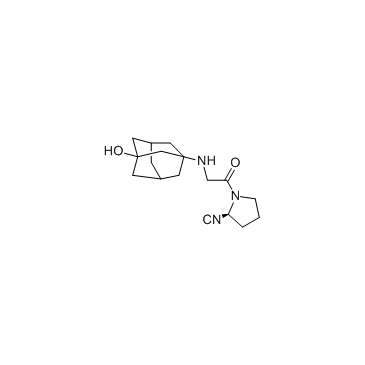 CAS#:274901-16-5
CAS#:274901-16-5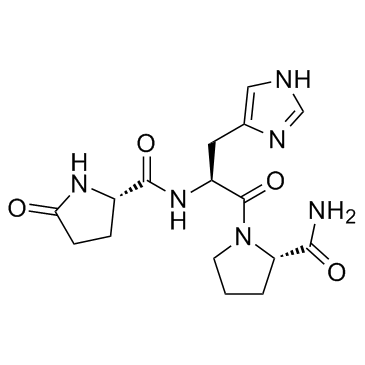 CAS#:24305-27-9
CAS#:24305-27-9 CAS#:207557-35-5
CAS#:207557-35-5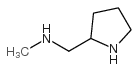 CAS#:66411-54-9
CAS#:66411-54-9
