Gramine
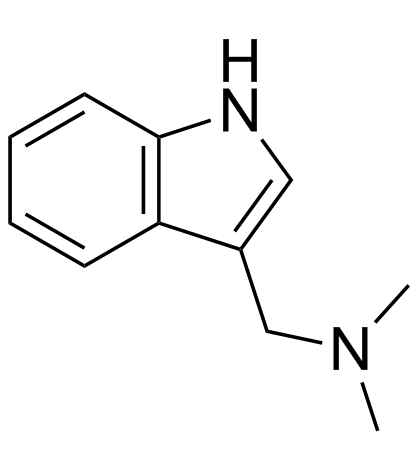
Gramine structure
|
Common Name | Gramine | ||
|---|---|---|---|---|
| CAS Number | 87-52-5 | Molecular Weight | 174.242 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 293.9±15.0 °C at 760 mmHg | |
| Molecular Formula | C11H14N2 | Melting Point | 132-134 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 131.5±20.4 °C | |
Use of GramineGramine (Donaxine) is a natural alkaloid isolated from giant reed[2], acts as an active adiponectin receptor (AdipoR) agonist, with IC50s of 3.2 and 4.2 µM for AdipoR2 and AdipoR1, respectively[1]. Gramine is also a human and mouse β2-Adrenergic receptor (β2-AR) agonist[2]. Gramine (Donaxine) has anti-tumor, anti-viral and anti-inflammatory properties[1]. |
| Name | 1-(1H-indol-3-yl)-N,N-dimethylmethanamine |
|---|---|
| Synonym | More Synonyms |
| Description | Gramine (Donaxine) is a natural alkaloid isolated from giant reed[2], acts as an active adiponectin receptor (AdipoR) agonist, with IC50s of 3.2 and 4.2 µM for AdipoR2 and AdipoR1, respectively[1]. Gramine is also a human and mouse β2-Adrenergic receptor (β2-AR) agonist[2]. Gramine (Donaxine) has anti-tumor, anti-viral and anti-inflammatory properties[1]. |
|---|---|
| Related Catalog | |
| Target |
IC50: 3.2 µM (AdipoR2), 4.2 µM (AdipoR1)[1] |
| In Vitro | Gramine is an active adiponectin receptor (AdipoR) agonist, with IC50s of 3.2 and 4.2 µM for AdipoR2 and AdipoR1, respectively[1]. Gramine is a potential β2-AR agonist[2]. Gramine (20 µM to 1.2 nM) dose-dependently inhibits the growth of AdipoR1/adipoR2-positive cancer cell lines (MDA-MB-231 and MCF-7 cells), with IC50s of 9.6±0.9 and 0.1±0.1 µM, respectively[1]. |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 293.9±15.0 °C at 760 mmHg |
| Melting Point | 132-134 °C(lit.) |
| Molecular Formula | C11H14N2 |
| Molecular Weight | 174.242 |
| Flash Point | 131.5±20.4 °C |
| Exact Mass | 174.115692 |
| PSA | 19.03000 |
| LogP | 1.90 |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.631 |
| InChIKey | OCDGBSUVYYVKQZ-UHFFFAOYSA-N |
| SMILES | CN(C)Cc1c[nH]c2ccccc12 |
| Storage condition | Refrigerator (+4°C) |
| Water Solubility | PRACTICALLY INSOLUBLE |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Hazard Codes | Xi:Irritant; |
|---|---|
| Risk Phrases | R36 |
| Safety Phrases | S45-S36/37/39-S26 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | NL7525000 |
| HS Code | 2933990090 |
| Precursor 9 | |
|---|---|
| DownStream 10 | |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
A retro-biosynthetic approach to the prediction of biosynthetic pathways from position-specific isotope analysis as shown for tramadol.
Proc. Natl. Acad. Sci. U. S. A. 112 , 8296-301, (2015) Tramadol, previously only known as a synthetic analgesic, has now been found in the bark and wood of roots of the African medicinal tree Nauclea latifolia. At present, no direct evidence is available ... |
|
|
NMR-titrations with complexes between ds-DNA and indole derivatives including tryptophane containing peptides.
FEBS Lett. 374(3) , 387-92, (1995) It is shown that NMR titrations can be used on a quantitative basis to derive binding constants and binding modes of ds-DNA ligand complexes from several signals. The results are partially at variance... |
|
|
Response of animals to dietary gramine. I. Performance and selected hematological, biochemical and histological parameters in growing chicken, rats and pigs.
Arch. Tierernahr. 55(1) , 1-16, (2001) The effects of feeding varied levels of low- and high-gramine yellow lupin seeds (LG and HG, respectively), and of synthetic gramine added to the diets in amounts ranging from 0.15 to 1.2 g per kg wer... |
|
Name: Primary qHTS for small molecule stabilizers of the endoplasmic reticulum resident pro...
Source: NCGC
Target: N/A
External Id: SERCaMPGLuc-p1-antagonist
|
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: QFRET-based biochemical primary high throughput screening assay to identify exosite i...
Source: The Scripps Research Institute Molecular Screening Center
Target: disintegrin and metalloproteinase domain-containing protein 17 preproprotein [Homo sapiens]
External Id: ADAM17_INH_QFRET_1536_1X%INH PRUN
|
|
Name: USP8 deubiquitinase inhibition: Primary qHTS
Source: 24642
Target: ubiquitin specific peptidase 8
External Id: USP8 FAST DUB HTS Primary
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: USP17 deubiquitinase inhibition: Primary qHTS
Source: 24642
Target: ubiquitin specific peptidase 17 like family member 5
External Id: USP17 FAST DUB HTS Primary
|
|
Name: USP7 deubiquitinase inhibition: Primary qHTS
Source: 24642
Target: ubiquitin specific peptidase 7
External Id: USP7 FAST DUB HTS Primary
|
|
Name: uHTS identification of small molecule activators of the adaptive arm of the Unfolded ...
Source: Burnham Center for Chemical Genomics
Target: N/A
External Id: BCCG-A405-UPR-XBP1-PrimaryAgonist-Assay
|
|
Name: A screen for compounds that inhibit the activity of LtaS in Staphylococcus aureus
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
External Id: HMS979
|
|
Name: Small Molecule Inhibitors of FGF22-Mediated Excitatory Synaptogenesis & Epilepsy Meas...
Source: Broad Institute
Target: FGF22 human recombinant
External Id: 7012-04_Inhibitor_Dose_CherryPick_Activity
|
| Gramin |
| Donaxine |
| Indole,3-[(dimethylamino)methyl] |
| Gramine |
| MFCD00005629 |
| 3-(Dimethylaminomethyl)indole |
| Indol-3-ylmethyldimethylamine |
| EINECS 201-749-8 |
| Donaxin |
| N,N-dimethyl-1H-indole-3-methanamine |
| 3-(N,N-dimethylaminomethyl) indole |
| N-(1H-indol-3-ylmethyl)-N,N-dimethylamine |
| 1H-Indole-3-methanamine,N,N-dimethyl |
| 1-(1H-Indol-3-yl)-N,N-dimethylmethanamine |
| 3-[(Dimethylamino)methyl]indole |
| (indol-3-ylmethyl)dimethylamine |
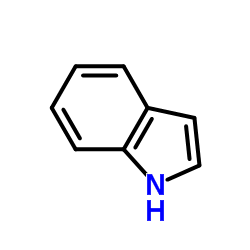 CAS#:120-72-9
CAS#:120-72-9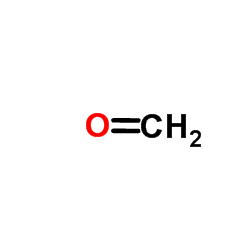 CAS#:50-00-0
CAS#:50-00-0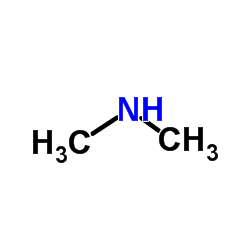 CAS#:124-40-3
CAS#:124-40-3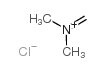 CAS#:30354-18-8
CAS#:30354-18-8 CAS#:51-80-9
CAS#:51-80-9 CAS#:27409-13-8
CAS#:27409-13-8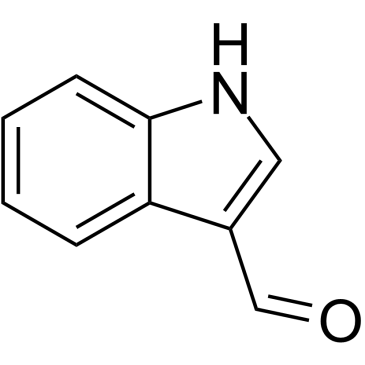 CAS#:487-89-8
CAS#:487-89-8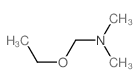 CAS#:62992-33-0
CAS#:62992-33-0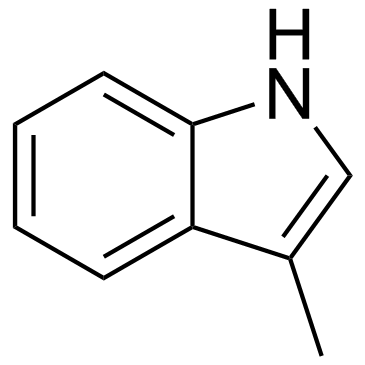 CAS#:83-34-1
CAS#:83-34-1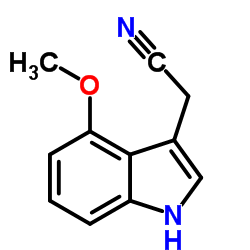 CAS#:4837-74-5
CAS#:4837-74-5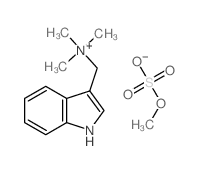 CAS#:19260-03-8
CAS#:19260-03-8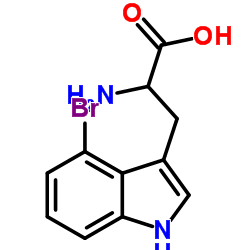 CAS#:25796-04-7
CAS#:25796-04-7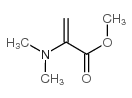 CAS#:999-59-7
CAS#:999-59-7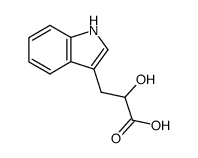 CAS#:832-97-3
CAS#:832-97-3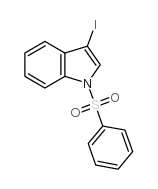 CAS#:80360-14-1
CAS#:80360-14-1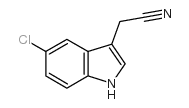 CAS#:81630-83-3
CAS#:81630-83-3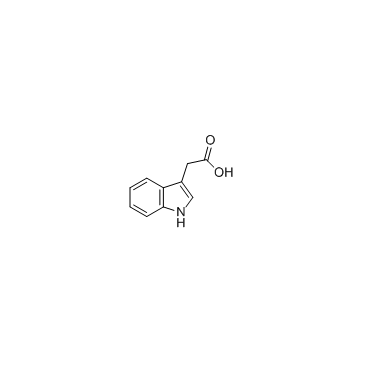 CAS#:87-51-4
CAS#:87-51-4
