Thiochrome
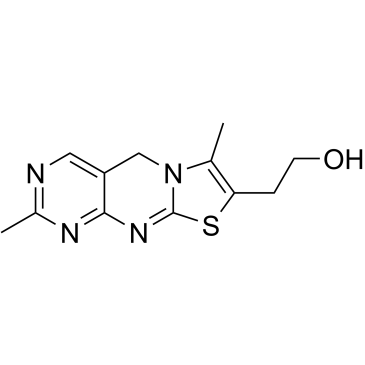
Thiochrome structure
|
Common Name | Thiochrome | ||
|---|---|---|---|---|
| CAS Number | 92-35-3 | Molecular Weight | 262.33100 | |
| Density | 1.49g/cm3 | Boiling Point | 462.6ºC at 760 mmHg | |
| Molecular Formula | C12H14N4OS | Melting Point | 228.8°C | |
| MSDS | Chinese USA | Flash Point | 233.5ºC | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of ThiochromeThiochrome, a natural oxidation product and metabolite of thiamine, is a selective M4 muscarinic receptor of acetylcholine (ACh) affinity enhancer. Thiochrome has neutral cooperativity with ACh at M1 to M3 receptors[1][2]. |
| Name | Thiochrome |
|---|---|
| Synonym | More Synonyms |
| Description | Thiochrome, a natural oxidation product and metabolite of thiamine, is a selective M4 muscarinic receptor of acetylcholine (ACh) affinity enhancer. Thiochrome has neutral cooperativity with ACh at M1 to M3 receptors[1][2]. |
|---|---|
| Related Catalog | |
| In Vivo | Thiochrome can increase the intensity of the reproduction process of the representatives of one-cell organisms worms, crustaceans, insects and fishes[1]. |
| References |
| Density | 1.49g/cm3 |
|---|---|
| Boiling Point | 462.6ºC at 760 mmHg |
| Melting Point | 228.8°C |
| Molecular Formula | C12H14N4OS |
| Molecular Weight | 262.33100 |
| Flash Point | 233.5ºC |
| Exact Mass | 262.08900 |
| PSA | 91.54000 |
| LogP | 0.52090 |
| Index of Refraction | 1.752 |
| InChIKey | GTQXMAIXVFLYKF-UHFFFAOYSA-N |
| SMILES | CC1=C(CCO)SC2=Nc3nc(C)ncc3CN21 |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26-S36 |
| RIDADR | NONH for all modes of transport |
| HS Code | 2934999090 |
| HS Code | 2934999090 |
|---|---|
| Summary | 2934999090. other heterocyclic compounds. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Functional activation of G-proteins coupled with muscarinic acetylcholine receptors in rat brain membranes.
J. Pharmacol. Sci. 125(2) , 157-68, (2014) The functional activation of Gi/o proteins coupled to muscarinic acetylcholine receptors (mAChRs) was investigated with the conventional guanosine-5'-O-(3-[(35)S]thio) triphosphate ([(35)S]GTPγS) bind... |
|
|
Salvage of the thiamin pyrimidine moiety by plant TenA proteins lacking an active-site cysteine.
Biochem. J. 463(1) , 145-55, (2014) The TenA protein family occurs in prokaryotes, plants and fungi; it has two subfamilies, one (TenA_C) having an active-site cysteine, the other (TenA_E) not. TenA_C proteins participate in thiamin sal... |
|
|
Determination of thiamine and its phosphate esters in rat tissues analyzed as thiochromes on a RP-amide C16 column.
J. Chromatogr. B. Analyt. Technol. Biomed. Life Sci. 816(1-2) , 67-72, (2005) A new reversed-phase chromatographic method is described for the separation and quantification of thiamine (T), thiamine monophosphate (TMP) and diphosphate (TDP) in rat tissues. Sample extraction wit... |
|
Name: Binding affinity to M4 receptor (unknown origin) assessed as fold change in presence ...
Source: ChEMBL
Target: Muscarinic acetylcholine receptor M4
External Id: CHEMBL5576405
|
|
Name: Human M1 receptor (Acetylcholine receptors (muscarinic))
Source: IUPHAR-DB
Target: M1 receptor (Acetylcholine receptors (muscarinic)) [Homo sapiens]
External Id: 13_Human
|
|
Name: Human M2 receptor (Acetylcholine receptors (muscarinic))
Source: IUPHAR-DB
Target: M2 receptor (Acetylcholine receptors (muscarinic)) [Homo sapiens]
External Id: 14_Human
|
|
Name: Human M3 receptor (Acetylcholine receptors (muscarinic))
Source: IUPHAR-DB
Target: M3 receptor (Acetylcholine receptors (muscarinic)) [Homo sapiens]
External Id: 15_Human
|
|
Name: Human M4 receptor (Acetylcholine receptors (muscarinic))
Source: IUPHAR-DB
Target: M4 receptor (Acetylcholine receptors (muscarinic)) [Homo sapiens]
External Id: 16_Human
|
| 2-(2,7-dimethyl-5H-pyrimido[4,5-d]thiazolo[3,2-a]pyrimidin-8-yl)-ethanol |
| 2-(2,7-Dimethyl-5H-pyrimido[4,5-d]thiazolo[3,2-a]pyrimidin-8-yl)-aethanol |
| 2-(2,7-dimethyl-5H-thiazolo[3',2':1,2]pyrimido[4,5-d]pyrimidin-8-yl)ethanol |
| 2-{6,12-dimethyl-4-thia-2,7,11,13-tetraazatricyclo[7.4.0.0^{3,7}]trideca-1(13),2,5,9,11-pentaen-5-yl}ethan-1-ol |
| 5H-Pyrimido(4,5-d)thiazolo(3,2-a)pyrimidine-8-ethanol,2,7-dimethyl |
| T7891_FLUKA |
| EINECS 202-149-9 |
| 2,7-dimethyl-5H-pyrimido[4,5-d]thiazolo[3,2-a]pyrimidine-8-ethanol |
| 2,7-Dimethylthiachromine-8-ethanol |
| THIOCHROME |
| Thiachrome |
| MFCD00010519 |
| TETRAKIS(2-ETHYLBUTYL)SILICATE |
 CAS#:67-03-8
CAS#:67-03-8 CAS#:70-16-6
CAS#:70-16-6 CAS#:13045-13-1
CAS#:13045-13-1![2,6a-dimethyl-6a,8,9,9a-tetrahydro-5H-furo[2',3':4,5]thiazolo[3,2-a]pyrimido[4,5-d]pyrimidine Structure](https://image.chemsrc.com/caspic/456/85196-67-4.png) CAS#:85196-67-4
CAS#:85196-67-4 CAS#:874-43-1
CAS#:874-43-1 CAS#:1124-01-2
CAS#:1124-01-2![N-[5-(2-imino-3a-methyl-tetrahydro-furo[2,3-d]thiazol-3-ylmethyl)-2-methyl-pyrimidin-4-yl]-formamide Structure](https://image.chemsrc.com/caspic/324/109044-67-9.png) CAS#:109044-67-9
CAS#:109044-67-9 CAS#:1153-39-5
CAS#:1153-39-5