U-69593
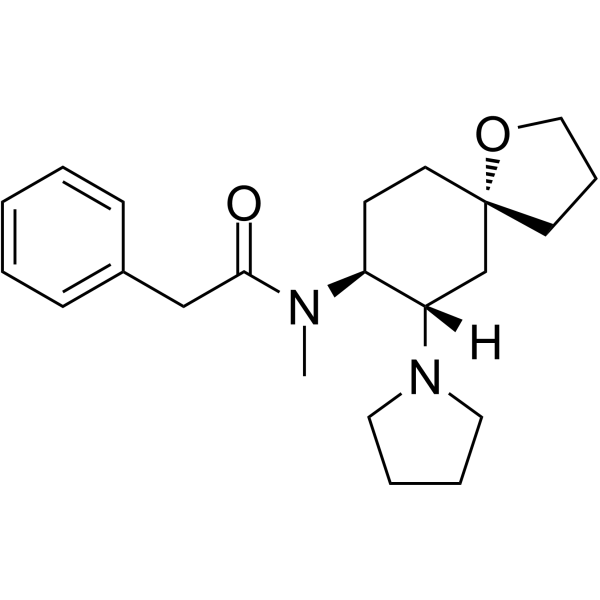
U-69593 structure
|
Common Name | U-69593 | ||
|---|---|---|---|---|
| CAS Number | 96744-75-1 | Molecular Weight | 356.50 | |
| Density | 1.14g/cm3 | Boiling Point | 518.719ºC at 760 mmHg | |
| Molecular Formula | C22H32N2O2 | Melting Point | N/A | |
| MSDS | USA | Flash Point | 267.512ºC | |
Use of U-69593U-69593 is a potent and selective κ1-opioid receptor agonist[1]. U-69593 attenuates cocaine-induced behavioral sensitization in the rat[2]. U-69593 reduces anxiety and enhances spontaneous alternation memory in mice[3]. U-69593 reduces calcium-dependent dialysate levels of dopamine and glutamate in the ventral striatum[4]. |
| Name | Tryptophan-P-1 |
|---|---|
| Synonym | More Synonyms |
| Description | U-69593 is a potent and selective κ1-opioid receptor agonist[1]. U-69593 attenuates cocaine-induced behavioral sensitization in the rat[2]. U-69593 reduces anxiety and enhances spontaneous alternation memory in mice[3]. U-69593 reduces calcium-dependent dialysate levels of dopamine and glutamate in the ventral striatum[4]. |
|---|---|
| Related Catalog | |
| In Vivo | U-69593 (0.16 mg/kg; s.c.) attenuates cocaine-induced behavioral sensitization in the rat[2]. U-69593 (1, 10, 25 nmol/µL; Microinjection) reduces anxiety and enhances spontaneous alternation memory in mice[3]. U-69593 (0.32 mg/kg; s.c.) decreases acute amphetamine-evoked behaviors and calcium-dependent dialysate levels of dopamine and glutamate in the ventral striatum[4]. Animal Model: Rat[2] Dosage: 0.16 mg/kg Administration: S.c. (an acute injection of cocaine (20 mg/kg i.p.)) Result: Attenuated the acute and chronic effects of cocaine on locomotor activity and stereotypy. Animal Model: CD-1 mice[3] Dosage: 1, 10, 25 nmol/µL Administration: Microinjection (in the infralimbic cortex (IL)), once every week, 2 weeks Result: Dose-dependently prolonged transfer-latency (T-L) and produced a dose-dependent anxiolytic behavioural profile, and after 24 h, the mouse were observed small but detectable carry-over effects. In week 2, U-69593 dose-dependently prolonged T/L and produced an anxiolytic behavioural profile in the first EPM (elevated plus-maze) trial, but observed a robust anxiolytic behavioural profile. Animal Model: 280-350 g, male Wistar rats[4] Dosage: 0.32 mg/kg Administration: S.c. (followed 15 min later by an injection of amphetamine (2.5 mg/kg i.p.)) Result: Significantly reduced the amphetamine-stimulated increase in dialysate dopamine levels and blocked the ability of amphetamine to evoke an increase in dialysate glutamate levels. |
| References |
| Density | 1.14g/cm3 |
|---|---|
| Boiling Point | 518.719ºC at 760 mmHg |
| Molecular Formula | C22H32N2O2 |
| Molecular Weight | 356.50 |
| Flash Point | 267.512ºC |
| Exact Mass | 356.24600 |
| PSA | 32.78000 |
| LogP | 3.19150 |
| Index of Refraction | 1.581 |
| InChIKey | PGZRDDYTKFZSFR-ONTIZHBOSA-N |
| SMILES | CN(C(=O)Cc1ccccc1)C1CCC2(CCCO2)CC1N1CCCC1 |
| Storage condition | 2-8°C |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | NONH for all modes of transport |
|
Comparison of liquid chromatography-mass spectrometry and radioimmunoassay for measurement of fentanyl and determination of pharmacokinetics in equine plasma.
J. Anal. Toxicol. 32(9) , 754-9, (2008) This study evaluated the validity of measuring fentanyl concentrations in equine plasma using radioimmunoassay (RIA) by comparing it to the established technique of liquid chromatography-mass spectrom... |
|
|
Physical presence of nor-binaltorphimine in mouse brain over 21 days after a single administration corresponds to its long-lasting antagonistic effect on κ-opioid receptors.
J. Pharmacol. Exp. Ther. 346(3) , 545-54, (2013) In the mouse 55°C warm-water tail-withdrawal assay, a single administration of nor-binaltorphimine (nor-BNI; 10 mg/kg i.p.) antagonized κ-opioid receptor (KOR) agonist-induced antinociception up to 14... |
|
|
Repeated stress dysregulates κ-opioid receptor signaling in the dorsal raphe through a p38α MAPK-dependent mechanism.
J. Neurosci. 32(36) , 12325-36, (2012) Repeated stress releases dynorphins and causes subsequent activation of κ-opioid receptors (KORs) in limbic brain regions. The serotonergic dorsal raphe nucleus (DRN) has previously been found to be a... |
| Trp-1 |
| Trytophan pyrolysate 1 |
| 1,4-Dimethyl-9H-pyrido(4,3-b)indol-3-amine |
| [3H]-Trp-P-1 |
| Tryptophan P1 |
| [14C]-Trp-P-1 |
| Trp-P-1 |
| 3-AMINO-1,4-DIMETHYL-5H-PYRIDO(4,3-B)INDOLE |
| U-69593 |