乙基硫脲
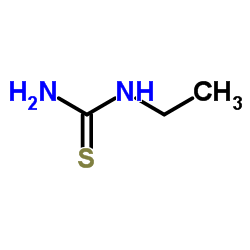
乙基硫脲结构式

|
常用名 | 乙基硫脲 | 英文名 | Ethylthiourea |
|---|---|---|---|---|
| CAS号 | 625-53-6 | 分子量 | 104.174 | |
| 密度 | 1.1±0.1 g/cm3 | 沸点 | 161.6±23.0 °C at 760 mmHg | |
| 分子式 | C3H8N2S | 熔点 | 108-110 °C(lit.) | |
| MSDS | 中文版 美版 | 闪点 | 51.5±22.6 °C | |
| 符号 |

GHS06 |
信号词 | Danger |
|
Structural and spectroscopic studies of some adducts of silver(I) halides with thiourea and N-ethyl substituted thioureas.
Dalton Trans. 39(18) , 4391-404, (2010) Syntheses, single crystal X-ray structural and spectroscopic characterizations are described for a variety of adducts of silver halides with thiourea ('tu'), N-ethylthiourea ('ettu' = EtNH.CS.NH(2)) and N,N'-diethylthiourea ('detu' = EtNH.CS.EtNH). This study... |
|
|
Infrared spectra of new Re(III) complexes with thiourea derivatives.
Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 58(14) , 3085-92, (2002) Complexes of the type [Re(III)L6]X3, with L = thiourea, N-methylthiourea, N-ethylthiourea or N,N'-dimethytlthiourea and X = Cl- or PF6-, were prepared as suitable precursors for the synthesis of new rhenium complexes potentially useful in nuclear medicine. Th... |
|
|
A convenient protocol for selective cleavage of 2-hydroxy acid amides. Application to semisynthesis of the cyclic heptapeptide aza HUN-7293.
J. Org. Chem. 67(24) , 8299-304, (2002) A two-step protocol for the first chemoselective cleavage of 2-hydroxy acid amides has been developed. Mesylation of the model substrate 2-(hydroxypropionylamino)-4-methylpentanoic acid methyl ester (11) followed by treatment with N-ethylthiourea (13) allows ... |
|
|
Endothelin-1 induces a glycolytic switch in pulmonary arterial endothelial cells via the mitochondrial translocation of endothelial nitric oxide synthase.
Am. J. Respir. Cell. Mol. Biol. 50(6) , 1084-95, (2014) Recent studies have indicated that, during the development of pulmonary hypertension (PH), there is a switch from oxidative phosphorylation to glycolysis in the pulmonary endothelium. However, the mechanisms underlying this phenomenon have not been elucidated... |
|
|
Synthesis and cytostatic properties of daunorubicin derivatives, containing N-phenylthiourea or N-ethylthiourea moieties in the 3'-position.
J. Antibiot. 44(2) , 192-9, (1991) A series of phenylthiourea and ethylthiourea derivatives of daunorubicin and its congeners was prepared by reaction of the 3'-amino group of the antibiotic with phenylisothiocyanate or ethylisothiocyanate. S-Methylation yielded S-methylisothiouromium salts wh... |