1-(3,5-BIS(TRIFLUOROMETHYL)PHENYL)-3-((1R)-(6-METHOXYQUINOLIN-4-YL)((2R,4S,5R)-5-VINYLQUINUCLIDIN-2-YL)METHYL)THIOUREA
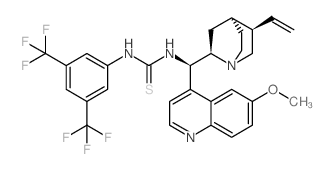
1-(3,5-BIS(TRIFLUOROMETHYL)PHENYL)-3-((1R)-(6-METHOXYQUINOLIN-4-YL)((2R,4S,5R)-5-VINYLQUINUCLIDIN-2-YL)METHYL)THIOUREA structure
|
Common Name | 1-(3,5-BIS(TRIFLUOROMETHYL)PHENYL)-3-((1R)-(6-METHOXYQUINOLIN-4-YL)((2R,4S,5R)-5-VINYLQUINUCLIDIN-2-YL)METHYL)THIOUREA | ||
|---|---|---|---|---|
| CAS Number | 852913-25-8 | Molecular Weight | 594.61400 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C29H28F6N4OS | Melting Point | 150°C | |
| MSDS | Chinese USA | Flash Point | N/A | |
| Symbol |

GHS06 |
Signal Word | Danger | |
|
Urea- and thiourea-substituted cinchona alkaloid derivatives as highly efficient bifunctional organocatalysts for the asymmetric addition of malonate to nitroalkenes: inversion of configuration at C9 dramatically improves catalyst performance.
Angew. Chem. Int. Ed. Engl. 44 , 6367-6370, (2005)
|
|
|
Highly enantioselective conjugate addition of nitromethane to chalcones using bifunctional cinchona organocatalysts.
Org. Lett. 7 , 1967-1969, (2005) Cinchona alkaloid-derived chiral bifunctional thiourea organocatalysts were synthesized and applied in the Michael addition between nitromethane and chalcones with high ee and chemical yields. |
Journals:
More...