nitromethane
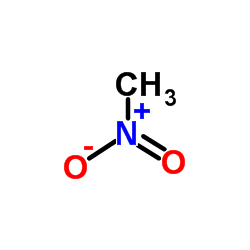
nitromethane structure
|
Common Name | nitromethane | ||
|---|---|---|---|---|
| CAS Number | 75-52-5 | Molecular Weight | 61.040 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 85.1±3.0 °C at 760 mmHg | |
| Molecular Formula | CH3NO2 | Melting Point | -29ºC | |
| MSDS | Chinese USA | Flash Point | 35.0±0.0 °C | |
| Symbol |



GHS02, GHS06, GHS08 |
Signal Word | Danger | |
|
Sensitive naked eye detection of hydrogen sulfide and nitric oxide by aza-BODIPY dyes in aqueous medium.
Anal. Chem. 86(18) , 9335-42, (2014) With an objective to develop optical probes for biologically important anions and neutral molecules, we synthesized three novel NIR absorbing aza-BODIPY derivatives, 3a-3c, and have systematically tuned their photophysical properties by changing the periphera... |
|
|
Generalized model of electromigration with 1:1 (analyte:selector) complexation stoichiometry: part II. Application to dual systems and experimental verification.
J. Chromatogr. A. 1384 , 147-54, (2015) Interactions among analyte forms that undergo simultaneous dissociation/protonation and complexation with multiple selectors take the shape of a highly interconnected multi-equilibrium scheme. This makes it difficult to express the effective mobility of the a... |
|
|
Determination of solubility parameters of ionic liquids and ionic liquid/solvent mixtures from intrinsic viscosity.
ChemPhysChem 15(16) , 3580-91, (2014) The total and partial solubility parameters (dispersion, polar and hydrogen-bonding solubility parameters) of ten ionic liquids were determined. Intrinsic viscosity approaches were used that encompassed a one-dimensional method (1D-Method), and two different ... |
|
|
Spectral assignment and proton transfer studies of N-(R-salicylidene)-1-amino-1-deoxy-D-sorbitols.
Magn. Reson. Chem. 53 , 849-52, (2015)
|
|
|
Deep Raman spectroscopy for the non-invasive standoff detection of concealed chemical threat agents.
Talanta 94 , 342-7, (2012) Deep Raman spectroscopy has been utilized for the standoff detection of concealed chemical threat agents from a distance of 15 m under real life background illumination conditions. By using combined time and space resolved measurements, various explosive prec... |
|
|
Highly stereoselective, cobalt(III)-directed Mannich additions in water yielding α-methylamino acid products.
Dalton Trans. 42(1) , 292-8, (2013) Highly stereoselective and rapid (<1 min) addition reactions to the imine double bond of 2-(methylimino)acetate complexes [L(4)Co(O(2)CCH=NCH(3))](2+) [L(4) = (en)(2) (7), (tren) (11)] were achieved in aqueous solution with nitromethane, ethyl 3-oxobutanoate ... |
|
|
Highly efficient enantioselective three-component synthesis of 2-amino-4H-chromenes catalysed by chiral tertiary amine-thioureas.
Chem. Commun. (Camb.) 48(47) , 5880-2, (2012) A three-component cascade reaction of salicylaldehyde, malononitrile/cyanoacetate and nitromethane catalysed by chiral tertiary amino-thioureas was developed, which leads to the production of highly functionalized 2-amino-4H-chromenes in good yields with good... |
|
|
Highly enantioselective conjugate addition of nitromethane to chalcones using bifunctional cinchona organocatalysts.
Org. Lett. 7 , 1967-1969, (2005) Cinchona alkaloid-derived chiral bifunctional thiourea organocatalysts were synthesized and applied in the Michael addition between nitromethane and chalcones with high ee and chemical yields. |
|
|
Reactions of atomic metal anions in the gas phase: competition between electron transfer, proton abstraction and bond activation.
J. Phys. Chem. A 115(48) , 14006-12, (2011) Bare metal anions K(-), Rb(-), Cs(-), Fe(-), Co(-), Ni(-), Cu(-), and Ag(-), generated by electrospray ionization of the corresponding oxalate or tricarballylate solutions, were allowed to react with methyl and ethyl chloride, methyl bromide, nitromethane, an... |
|
|
Solvent-minimized extraction for determining halonitromethanes and trihalomethanes in water
J. Chromatogr. A. 1248 , 1-8, (2012) Highlights ► A MLLE method has been developed for the whole array determination of HNMs and THMs. ► The water stability is obtained by its acidification at the time of collection. ► LVI using solvent vent mode enhances the sensitivity of the MLLE method. |