| Structure | Name/CAS No. | Articles |
|---|---|---|
 |
Sulfapyridine
CAS:144-83-2 |
|
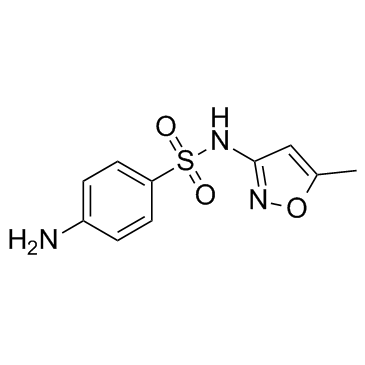 |
Sulfamethoxazole
CAS:723-46-6 |
|
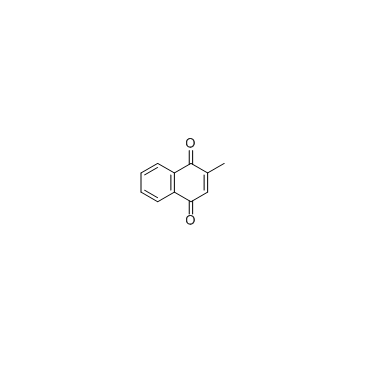 |
Menadione
CAS:58-27-5 |
|
 |
Sulfathiazole
CAS:72-14-0 |
|
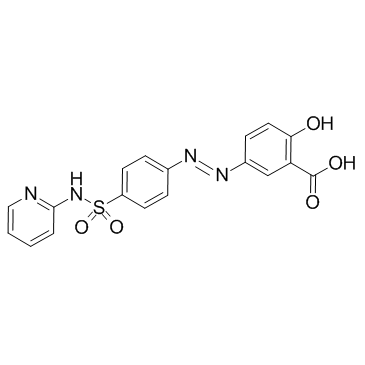 |
sulfasalazine
CAS:599-79-1 |
|
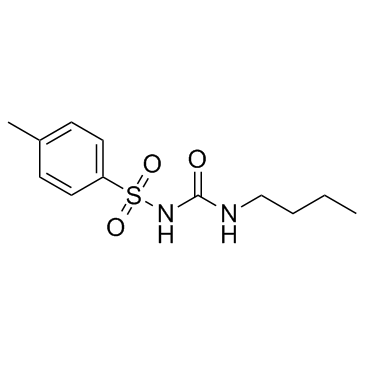 |
Tolbutamide
CAS:64-77-7 |
|
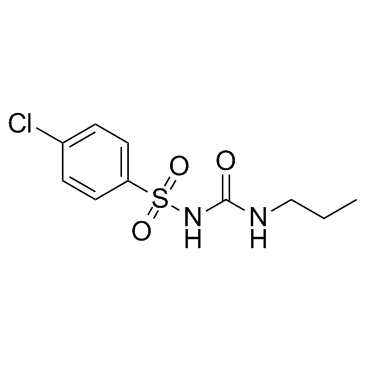 |
chlorpropamide
CAS:94-20-2 |
|
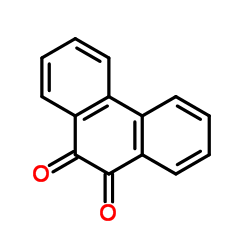 |
Phenanthrene-9,10-dione
CAS:84-11-7 |
|
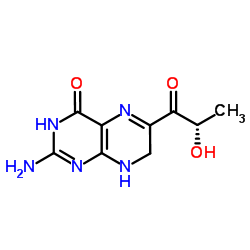 |
L-Sepiapterin
CAS:17094-01-8 |