Inosine,6-S-methyl-6-thio
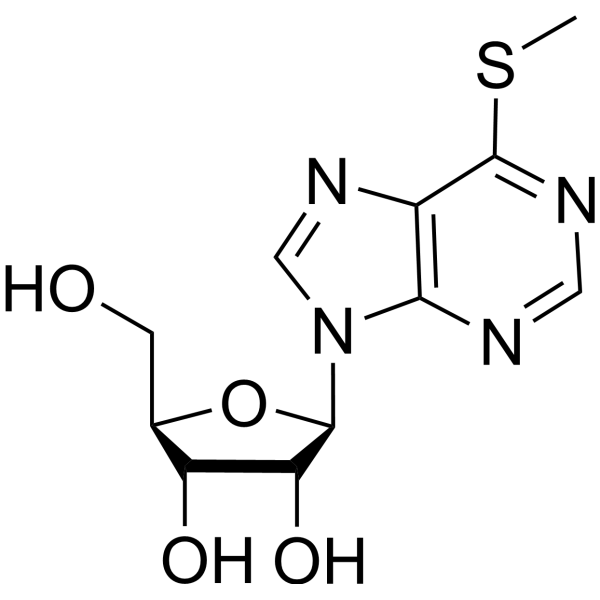
Inosine,6-S-methyl-6-thio structure
|
Common Name | Inosine,6-S-methyl-6-thio | ||
|---|---|---|---|---|
| CAS Number | 342-69-8 | Molecular Weight | 298.32 | |
| Density | 1.85g/cm3 | Boiling Point | 636.3ºC at 760mmHg | |
| Molecular Formula | C11H14N4O4S | Melting Point | 166°C | |
| MSDS | Chinese USA | Flash Point | 338.6ºC | |
Use of Inosine,6-S-methyl-6-thio6-Methylmercaptopurine riboside is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc[1]. |
| Name | 6-methylthioinosine |
|---|---|
| Synonym | More Synonyms |
| Description | 6-Methylmercaptopurine riboside is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.85g/cm3 |
|---|---|
| Boiling Point | 636.3ºC at 760mmHg |
| Melting Point | 166°C |
| Molecular Formula | C11H14N4O4S |
| Molecular Weight | 298.32 |
| Flash Point | 338.6ºC |
| Exact Mass | 298.07400 |
| PSA | 138.82000 |
| Index of Refraction | 1.822 |
| InChIKey | ZDRFDHHANOYUTE-IOSLPCCCSA-N |
| SMILES | CSc1ncnc2c1ncn2C1OC(CO)C(O)C1O |
| Storage condition | −20°C |
| Stability | Incompatible with strong oxidizing agents. |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Safety Phrases | S22-S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | UO8985000 |
| HS Code | 29349990 |
| Precursor 9 | |
|---|---|
| DownStream 9 | |
|
The role of inosine-5'-monophosphate dehydrogenase in thiopurine metabolism in patients with inflammatory bowel disease.
Ther. Drug Monit. 33 , 200-208, (2011) There is a large interindividual variability in thiopurine metabolism. High concentrations of methylthioinosine-5'-monophosphate (meTIMP) and low concentrations of 6-thioguanine nucleotides (6-TGNs) h... |
|
|
Methylthioadenosine deaminase in an alternative quorum sensing pathway in Pseudomonas aeruginosa.
Biochemistry 51(45) , 9094-103, (2012) Pseudomonas aeruginosa possesses an unusual pathway for 5'-methylthioadenosine (MTA) metabolism involving deamination to 5'-methylthioinosine (MTI) followed by N-ribosyl phosphorolysis to hypoxanthine... |
|
|
A prospective, open-label trial of 6-thioguanine in patients with ulcerative or indeterminate colitis.
Scand. J. Gastroenterol. 40(10) , 1205-13, (2005) 6-thioguanine (6-TG) has emerged as a promising therapeutic alternative in patients with Crohn's disease intolerant or resistant to azathioprine (AZA) and/or 6-mercaptopurine (6-MP). The aim of the pr... |
|
Name: Concentration required to inhibit HSV type 1 strain HF induced cytopathogenic affects...
Source: ChEMBL
Target: Human alphaherpesvirus 1
External Id: CHEMBL694222
|
|
Name: Virus rating against rhinovirus type 1A
Source: ChEMBL
Target: Human rhinovirus 1A
External Id: CHEMBL800916
|
|
Name: Virus rating of against herpes simplex virus type 2
Source: ChEMBL
Target: Human alphaherpesvirus 2
External Id: CHEMBL696293
|
|
Name: Antiviral activity against Zika virus PE243 infected in African green monkey Vero cel...
Source: ChEMBL
Target: Zika virus
External Id: CHEMBL4337947
|
|
Name: In vitro antiviral activity in secondary cultures of rabbit kidney cells infected wit...
Source: ChEMBL
Target: Human alphaherpesvirus 1
External Id: CHEMBL877527
|
|
Name: Virus rating against parainfluenza virus type 3
Source: ChEMBL
Target: Human respirovirus 3
External Id: CHEMBL762072
|
|
Name: Inhibition Eimeria tenella growth
Source: ChEMBL
Target: Eimeria tenella
External Id: CHEMBL674773
|
|
Name: Antiviral activity against SARS-CoV Frankfurt1 in Vero E6 cells by plaque reduction a...
Source: ChEMBL
Target: Severe acute respiratory syndrome-related coronavirus
External Id: CHEMBL887577
|
|
Name: Inhibition of Cellular Replication of friend erythroleukemia cells
Source: ChEMBL
Target: Erythroleukemia cell line
External Id: CHEMBL681343
|
|
Name: Virus rating against adenovirus type 2
Source: ChEMBL
Target: Human adenovirus 2
External Id: CHEMBL641423
|
| EINECS 206-442-2 |
| 6-MethylmercaptopurineRiboside |
| MFCD00022826 |
| 6-Methylthioinosine |
| METHYLTHIOINOSINE |
| 6-Methylmercaptopurinribosid |
| 6-methylmp-riboside |
| 6-MMPR |
| 6-METHYLTHIOPURINE RIBOSIDE |
| 6-Methylmercaptopurine riboside |
 CAS#:5188-07-8
CAS#:5188-07-8 CAS#:7387-57-7
CAS#:7387-57-7 CAS#:58-61-7
CAS#:58-61-7 CAS#:163632-54-0
CAS#:163632-54-0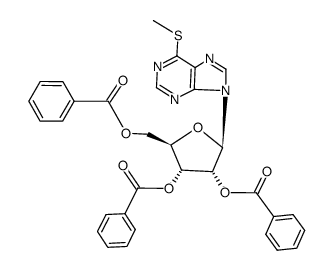 CAS#:66782-10-3
CAS#:66782-10-3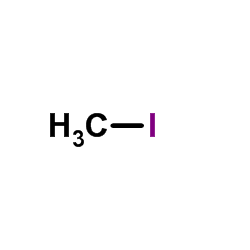 CAS#:74-88-4
CAS#:74-88-4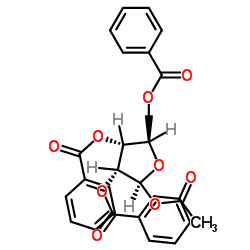 CAS#:6974-32-9
CAS#:6974-32-9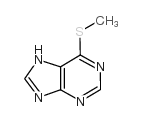 CAS#:50-66-8
CAS#:50-66-8 CAS#:73237-86-2
CAS#:73237-86-2 CAS#:18646-11-2
CAS#:18646-11-2 CAS#:4338-47-0
CAS#:4338-47-0 CAS#:23526-11-6
CAS#:23526-11-6![[7,7-dimethyl-2-(6-methylsulfanylpurin-9-yl)-3,6,8-trioxabicyclo[3.3.0]oct-4-yl]methanol structure](https://image.chemsrc.com/caspic/259/42021-78-3.png) CAS#:42021-78-3
CAS#:42021-78-3 CAS#:132154-36-0
CAS#:132154-36-0 CAS#:4542-23-8
CAS#:4542-23-8 CAS#:53665-01-3
CAS#:53665-01-3