Sodium oxamate
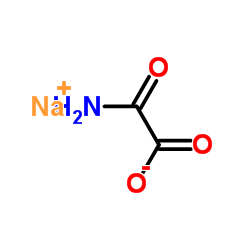
Sodium oxamate structure
|
Common Name | Sodium oxamate | ||
|---|---|---|---|---|
| CAS Number | 565-73-1 | Molecular Weight | 111.032 | |
| Density | N/A | Boiling Point | 306.3ºC at 760 mmHg | |
| Molecular Formula | C2H2NNaO3 | Melting Point | 300 °C | |
| MSDS | Chinese USA | Flash Point | 139ºC | |
Use of Sodium oxamateOxamic acid (oxamate) sodium salt is a lactate dehydrogenase-A (LDH-A) inhibitor. Oxamic acid sodium salt shows anti-tumor activity, and anti-proliferative activity against cancer cells, and can induce apoptosis[1][2][3]. |
| Name | oxamic acid sodium salt |
|---|---|
| Synonym | More Synonyms |
| Description | Oxamic acid (oxamate) sodium salt is a lactate dehydrogenase-A (LDH-A) inhibitor. Oxamic acid sodium salt shows anti-tumor activity, and anti-proliferative activity against cancer cells, and can induce apoptosis[1][2][3]. |
|---|---|
| Related Catalog | |
| In Vitro | Oxamic acid suppresses the proliferation, migration and invasion of both A2780 and SKOV3 cells[1]. Oxamic acid (10 μM; 24-72 h) inhibits cell proliferation in a dose- and time-dependent manner in both NPC cancer cells[2]. Oxamic acid (0-100 mM; 24 h) induces cell cycle arrest in the G2/M phase in CNE-1 and CNE-2 cells[2]. Oxamic acid (0-100 mM; 48 h) induces apoptosis via caspase-3 activation and the mitochondrial pathway in NPC cells[2]. Oxamic acid (0-100 mM; 24 h) increases ROS levels in NPC cells[2]. Cell Proliferation Assay[2] Cell Line: CNE-1 and CNE-2 cells Concentration: 10 μM Incubation Time: 24-72 hours Result: Showed IC50s of 74.6, 32.4 and 17.8 mM and 62.3, 44.5, 31.6 mM at 24, 48 and 72 h in the CNE-1 and CNE-2 cancer cells, respectively. Apoptosis Analysis[2] Cell Line: 0, 20, 50 and 100 mM Concentration: 10 μM Incubation Time: 48 hours Result: Showed the increasement of early and late apoptotic cells in a dose-dependent manner. Increased the expression of pro-apoptotic Bax and cleaved-caspase-3, while reduced the anti-apoptotic signals of Bcl-2 and pro-caspase-3. Cell Cycle Analysis[2] Cell Line: 0, 20, 50 and 100 mM Concentration: 10 μM Incubation Time: 24 hours Result: Showed a dose-dependent increase in the numbers of CNE-1 and CNE-2 cells in the G2/M phase. |
| In Vivo | Oxamic acid (intraperitoneal injection; 750 mg/kg; once daily; 3 w) treatment improves the efficacy of tumor inhibition in vivo when combined with irradiation treatment[2]. Animal Model: Female Balb/c nude mice injected with CNE-1 cells[2] Dosage: 750 mg/kg Administration: Intraperitoneal injection; 750 mg/kg; once daily; 3 weeks Result: Inhibited the tumor growth when compared to either oxamate alone or irradiation alone. |
| Boiling Point | 306.3ºC at 760 mmHg |
|---|---|
| Melting Point | 300 °C |
| Molecular Formula | C2H2NNaO3 |
| Molecular Weight | 111.032 |
| Flash Point | 139ºC |
| Exact Mass | 110.993240 |
| PSA | 83.22000 |
| InChIKey | RQVZIJIQDCGIKI-UHFFFAOYSA-M |
| SMILES | NC(=O)C(=O)[O-].[Na+] |
| Storage condition | Store at RT. |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | F: Flammable; |
| Risk Phrases | R22;R24/25 |
| Safety Phrases | S22-S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 2924199090 |
| HS Code | 2924199090 |
|---|---|
| Summary | 2924199090. other acyclic amides (including acyclic carbamates) and their derivatives; salts thereof. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Targeting metabolic flexibility by simultaneously inhibiting respiratory complex I and lactate generation retards melanoma progression.
Oncotarget 6 , 37281-99, (2015) Melanoma is a largely incurable skin malignancy owing to the underlying molecular and metabolic heterogeneity confounded by the development of resistance. Cancer cells have metabolic flexibility in ch... |
|
|
Chemical genetics reveals a complex functional ground state of neural stem cells.
Nat. Chem. Biol. 3(5) , 268-273, (2007) The identification of self-renewing and multipotent neural stem cells (NSCs) in the mammalian brain holds promise for the treatment of neurological diseases and has yielded new insight into brain canc... |
|
|
Different effects of LDH-A inhibition by oxamate in non-small cell lung cancer cells.
Oncotarget 5(23) , 11886-96, (2015) Higher rate of glycolysis has been long observed in cancer cells, as a vital enzyme in glycolysis, lactate dehydrogenase A (LDH-A) has been shown with great potential as an anti-cancer target. Accumul... |
|
Name: Inhibition of neurosphere proliferation of mouse neural precursor cells by MTT assay
Source: ChEMBL
Target: N/A
External Id: CHEMBL1266185
|
|
Name: Small-molecule inhibitors of ST2 (IL1RL1)
Source: 20881
Target: interleukin-1 receptor-like 1 isoform [homo sapiens]
External Id: ST2_IL33_Inhibitors_Primary_Screening_77700
|
|
Name: Rescue cell viability in cybrid cells with a genetic mutation in complex 1 of the mit...
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
Target: N/A
External Id: HMS1315
|
|
Name: qHTS assay for measurement of the activity of small molecule inhibitors on Zika virus...
Source: NCGC
Target: N/A
External Id: ZIK097
|
|
Name: 96-well format Chlamydomonas reinhardtii Algae Gravitaxis Assay to measure the differ...
Source: University of Pittsburgh Molecular Library Screening Center
Target: N/A
External Id: MH081217, Chlamydomonas reinhardtii Gravitaxis Assay to Identify Small Molecule Inhibitors of Cilia.
|
|
Name: Dicer-mediated maturation of pre-microRNA
Source: Center for Chemical Genomics, University of Michigan
Target: N/A
External Id: TargetID_659_CEMA
|
|
Name: Competitive inhibition of human LDH5 in presence of NADH
Source: ChEMBL
Target: L-lactate dehydrogenase A chain
External Id: CHEMBL3768756
|
|
Name: High-throughput screen for inhibitors of the GIV GBA-motif interaction with Galpha-i ...
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
External Id: HMS1303
|
|
Name: Inhibition of recombinant human full-length N-terminal His-tagged LDHA (1 to 332 resi...
Source: ChEMBL
Target: L-lactate dehydrogenase A chain
External Id: CHEMBL4396270
|
|
Name: qHTS assay to identify small molecule antagonists of the estrogen related receptor (E...
Source: 824
Target: N/A
External Id: ERR513
|
| Sodium oxamate |
| MFCD00044553 |
| sodium,oxamate |
| Acetic acid, 2-amino-2-oxo-, sodium salt (1:1) |
| Sodium amino(oxo)acetate |
| Oxalic acid monoamide sodium salt |
| Acetic acid, aminooxo-, monosodium salt |
| EINECS 209-290-5 |
| OXAMIC ACID SODIUM SALT |

