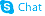Chlorobutanol hemihydrate
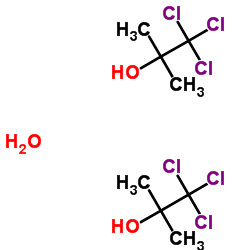
Chlorobutanol hemihydrate structure
|
Common Name | Chlorobutanol hemihydrate | ||
|---|---|---|---|---|
| CAS Number | 6001-64-5 | Molecular Weight | 372.929 | |
| Density | N/A | Boiling Point | 167ºC | |
| Molecular Formula | C4H9Cl3O2 | Melting Point | 75-79ºC | |
| MSDS | Chinese USA | Flash Point | 100ºC | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of Chlorobutanol hemihydrateChlorobutanol hemihydrate is a pharmaceutical preservative with sedative-hypnotic actions. Chlorobutanol hemihydrate is active against a wide variety of Gram-positive and Gram-negative bacteria, and several mold spores and fungi. Chlorobutanol hemihydrate is widely used in food and cosmetic industry[1][2]. |
| Name | 1,1,1-Trichloro-2-Methyl-2-Propanol Hemihydrate |
|---|---|
| Synonym | More Synonyms |
| Description | Chlorobutanol hemihydrate is a pharmaceutical preservative with sedative-hypnotic actions. Chlorobutanol hemihydrate is active against a wide variety of Gram-positive and Gram-negative bacteria, and several mold spores and fungi. Chlorobutanol hemihydrate is widely used in food and cosmetic industry[1][2]. |
|---|---|
| Related Catalog | |
| References |
| Boiling Point | 167ºC |
|---|---|
| Melting Point | 75-79ºC |
| Molecular Formula | C4H9Cl3O2 |
| Molecular Weight | 372.929 |
| Flash Point | 100ºC |
| Exact Mass | 369.923065 |
| PSA | 29.46000 |
| LogP | 2.06320 |
| Appearance of Characters | Crystalline Powder, Crystals and/or Chunks | White to almost white |
| Storage condition | -20°C |
| Stability | Stable. Generates toxic fumes on combustion. Incompatible with strong oxidizing agents. |
| Water Solubility | Slightly soluble in water, very soluble in ethanol (96 per cent), soluble in glycerol (85 per cent). | Soluble in ethanol, ether, chloroform, and glycerol. Insoluble in water. |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302 |
| Precautionary Statements | P301 + P312 + P330 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn:Harmful; |
| Risk Phrases | R22;R36/37/38 |
| Safety Phrases | 26-36/37/39 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | UC0175000 |
|
An efficient synthesis of a dual PPAR alpha/gamma agonist and the formation of a sterically congested alpha-aryloxyisobutyric acid via a Bargellini reaction.
J. Org. Chem. 70(21) , 8560-3, (2005) A practical synthesis of benzisoxazole 1 and its conversion to alpha-aryloxyisobutyric acid 2 using 1,1,1-trichloro-2-methyl-2-propanol (chloretone) was developed. Benzisoxazole 1 was formed in high y... |
|
|
Application of a capillary electrophoresis method for simultaneous determination of preservatives in pharmaceutical formulations.
J. Sep. Sci. 28(2) , 137-43, (2005) Preservatives are used to protect pharmaceutical formulations from microbial attack during the period of administration to the patient. Because of their biological activity, preservatives have to be i... |
|
|
Freeze-drying above room temperature.
J. Pharm. Sci. 88(5) , 501-6, (1999) This study investigates the use of solid, organic compounds to lyophilize drugs without conventional freeze-drying equipment. The aim of the investigation is to find a pharmaceutically acceptable solv... |
| 1,1,1-Trichloro-2-methyl-2-propanol hemihydrate |
| 1,1,1-Trichloro-2-methyl-2-propanol hydrate (2:1) |
| MFCD02179352 |
| 1,1,1-trichloro-2-methyl-2-propanol(hydrate) (2:1) |
| Chlorobutanol Hemihydrate |
| 1,1,1-Trichloro-2-methylpropan-2-ol hydrate (2:1) |
| 2-Propanol, 1,1,1-trichloro-2-methyl-, hydrate (2:1) |