5-Aminovaleric acid
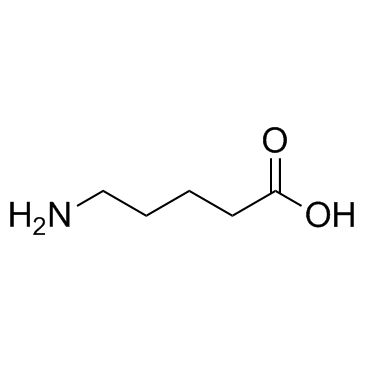
5-Aminovaleric acid structure
|
Common Name | 5-Aminovaleric acid | ||
|---|---|---|---|---|
| CAS Number | 660-88-8 | Molecular Weight | 117.146 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 247.5±23.0 °C at 760 mmHg | |
| Molecular Formula | C5H11NO2 | Melting Point | 158-161 °C(lit.) | |
| MSDS | USA | Flash Point | 103.5±22.6 °C | |
Use of 5-Aminovaleric acid5-Aminovaleric acid is believed to act as a methylene homologue of gamma-aminobutyric acid (GABA) and functions as a weak GABA agonist. |
| Name | 5-aminopentanoic acid |
|---|---|
| Synonym | More Synonyms |
| Description | 5-Aminovaleric acid is believed to act as a methylene homologue of gamma-aminobutyric acid (GABA) and functions as a weak GABA agonist. |
|---|---|
| Related Catalog | |
| Target |
Human Endogenous Metabolite |
| In Vitro | 5-Aminovaleric acid is a normal metabolite present in human saliva, with a tendency to elevated concentration in patients with chronic periodontitis[1]. 5-Aminovaleric acid is also believed to act as a methylene homologue of gamma-aminobutyric acid (GABA) and functions as a weak GABA agonist[2]. It is also known as an antifibrinolytic amino acid analog and so it functions as a weak inhibitor of the blood clotting pathway[3]. |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 247.5±23.0 °C at 760 mmHg |
| Melting Point | 158-161 °C(lit.) |
| Molecular Formula | C5H11NO2 |
| Molecular Weight | 117.146 |
| Flash Point | 103.5±22.6 °C |
| Exact Mass | 117.078979 |
| PSA | 63.32000 |
| LogP | -0.34 |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.466 |
| Storage condition | Store at 0-5°C |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xn |
| Risk Phrases | R20/21/22 |
| Safety Phrases | S22-S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 2922499990 |
| HS Code | 2922499990 |
|---|---|
| Summary | HS:2922499990 other amino-acids, other than those containing more than one kind of oxygen function, and their esters; salts thereof VAT:17.0% Tax rebate rate:9.0% Supervision conditions:AB(certificate of inspection for goods inward,certificate of inspection for goods outward) MFN tariff:6.5% General tariff:30.0% |
|
Peptide dimethylation: fragmentation control via distancing the dimethylamino group.
J. Am. Soc. Mass Spectrom. 25(10) , 1694-704, (2014) Direct reductive methylation of peptides is a common method for quantitative proteomics. It is an active derivatization technique; with participation of the dimethylamino group, the derivatized peptid... |
|
|
Detection of autosomal dominant polycystic kidney disease by NMR spectroscopic fingerprinting of urine.
Kidney Int. 79(11) , 1244-53, (2011) Autosomal dominant polycystic kidney disease (ADPKD) is a frequent cause of kidney failure; however, urinary biomarkers for the disease are lacking. In a step towards identifying such markers, we used... |
|
|
Highly sensitive GC/MS/MS method for quantitation of amino and nonamino organic acids.
Anal. Chem. 83(7) , 2705-11, (2011) Metabolite profiling methods are important tools for measurement of metabolite pools in biological systems. While most metabolite profiling methods report relative intensities or depend on a few inter... |
| 5-amino valeric acid |
| δ-Aminovaleric acid |
| EINECS 211-544-5 |
| 5-Aminopentanoic acid |
| 5-Aminovaleric acid |
| MFCD00008232 |
| H-5-Ava-OH |

