Phenindione
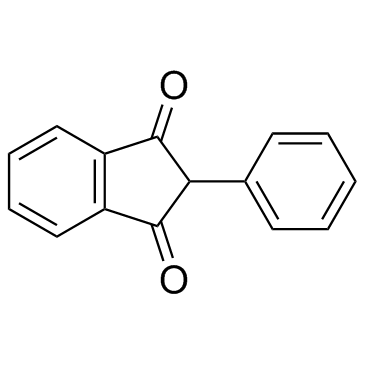
Phenindione structure
|
Common Name | Phenindione | ||
|---|---|---|---|---|
| CAS Number | 83-12-5 | Molecular Weight | 222.23900 | |
| Density | 1.343g/cm3 | Boiling Point | 243.3 °C (0.3 mmHg) | |
| Molecular Formula | C15H10O2 | Melting Point | 144-148 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 208 °C | |
| Symbol |

GHS06 |
Signal Word | Danger | |
Use of PhenindionePhenindione is an anticoagulant which functions as a Vitamin K antagonist.Target: OthersPhenindione(Rectadione) is an anticoagulant which functions as a Vitamin K antagonist. A lymphocyte transformation test showed proliferation of T-cells from the hypersensitive patient, but not from four controls on exposure to phenindione in vitro. Drug-specific T-cell clones were generated and characterized in terms of their phenotype, functionality, and mechanism of antigen presentation. Forty-three human leukocyte antigen class II restricted CD4+ αβ T-cell clones were identified. T-cell activation resulted in the secretion of interferon-γ and interleukin-5 [1]. |
| Name | phenindione |
|---|---|
| Synonym | More Synonyms |
| Description | Phenindione is an anticoagulant which functions as a Vitamin K antagonist.Target: OthersPhenindione(Rectadione) is an anticoagulant which functions as a Vitamin K antagonist. A lymphocyte transformation test showed proliferation of T-cells from the hypersensitive patient, but not from four controls on exposure to phenindione in vitro. Drug-specific T-cell clones were generated and characterized in terms of their phenotype, functionality, and mechanism of antigen presentation. Forty-three human leukocyte antigen class II restricted CD4+ αβ T-cell clones were identified. T-cell activation resulted in the secretion of interferon-γ and interleukin-5 [1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.343g/cm3 |
|---|---|
| Boiling Point | 243.3 °C (0.3 mmHg) |
| Melting Point | 144-148 °C(lit.) |
| Molecular Formula | C15H10O2 |
| Molecular Weight | 222.23900 |
| Flash Point | 208 °C |
| Exact Mass | 222.06800 |
| PSA | 34.14000 |
| LogP | 2.84940 |
| Index of Refraction | 1.703 |
| InChIKey | NFBAXHOPROOJAW-UHFFFAOYSA-N |
| SMILES | O=C1c2ccccc2C(=O)C1c1ccccc1 |
| Storage condition | 2-8°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H301-H317 |
| Precautionary Statements | P280-P301 + P310 |
| Personal Protective Equipment | Eyeshields;Faceshields;Gloves;type P2 (EN 143) respirator cartridges |
| Hazard Codes | T: Toxic; |
| Risk Phrases | R25 |
| Safety Phrases | S36/37/39-S45 |
| RIDADR | UN 2811 6.1/PG 3 |
| WGK Germany | 3 |
| RTECS | NK6125000 |
| Packaging Group | III |
| Hazard Class | 6.1(b) |
| HS Code | 2914399090 |
| Precursor 8 | |
|---|---|
| DownStream 10 | |
| HS Code | 2914399090 |
|---|---|
| Summary | 2914399090. other aromatic ketones without other oxygen function. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:5.5%. General tariff:30.0% |
|
Multi-target spectral moment QSAR versus ANN for antiparasitic drugs against different parasite species.
Bioorg. Med. Chem. 18 , 2225-31, (2010) There are many of pathogen parasite species with different susceptibility profile to antiparasitic drugs. Unfortunately, almost QSAR models predict the biological activity of drugs against only one pa... |
|
|
Development of a phospholipidosis database and predictive quantitative structure-activity relationship (QSAR) models.
Toxicol. Mech. Methods 18 , 217-27, (2008) ABSTRACT Drug-induced phospholipidosis (PL) is a condition characterized by the accumulation of phospholipids and drug in lysosomes, and is found in a variety of tissue types. PL is frequently manifes... |
|
|
Maintenance of anticoagulant and antiplatelet agents for patients undergoing peribulbar anesthesia and vitreoretinal surgery.
Retina (Philadelphia, Pa.) 32(9) , 1868-73, (2012) To establish the prevalence of anticoagulation (vitamin K antagonists) and antiplatelet agent therapy in patients undergoing vitreoretinal surgery and to compare the outcome of peribulbar anesthesia a... |
|
Name: Primary qHTS assay for inhibitors of alpha-synuclein gene (SNCA) expression
Source: NCGC
External Id: SNCA-p-activity-luciferase
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Cytochrome P450 Family 1 Subfamily A Member 2 (CYP1A2) small molecule antagonists: lu...
Source: 824
External Id: CYP273
|
|
Name: Increase the activity of the Burkholderia fixLJ 2-component system
Source: ICCB-Longwood/NSRB Screening Facility, Harvard Medical School
Target: Burkholderia multivorans
External Id: HMS1625
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: qHTS Assay for Small Molecule Inhibitors of the Human hERG Channel Activity
Source: NCGC
External Id: HERG01
|
|
Name: qHTS for Inhibitors of TGF-b: Cytotox Counterscreen
Source: NCGC
Target: N/A
External Id: SMAD3201
|
|
Name: uHTS identification of cystic fibrosis induced NFkb Inhibitors in a fluoresence assay
Source: Burnham Center for Chemical Genomics
Target: cystic fibrosis transmembrane conductance regulator [Homo sapiens]
External Id: SBCCG-A764-CF-PAF-Primary-Assay
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ant...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_ANT_FLUO8_1536_1X%INH PRUN
|
| 2-PHENYL-1,3-INDANDIONE |
| 2-phenylindene-1,3-dione |
| EINECS 201-454-4 |
| MFCD00003782 |
 CAS#:74942-26-0
CAS#:74942-26-0 CAS#:151-88-2
CAS#:151-88-2 CAS#:1807-49-4
CAS#:1807-49-4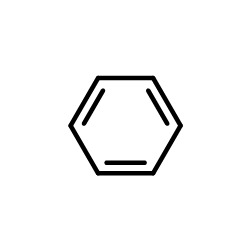 CAS#:71-43-2
CAS#:71-43-2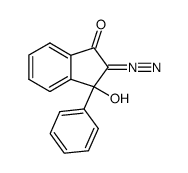 CAS#:79963-47-6
CAS#:79963-47-6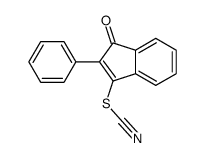 CAS#:91512-16-2
CAS#:91512-16-2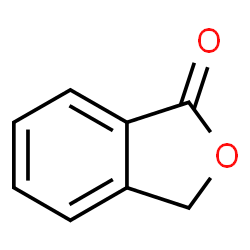 CAS#:87-41-2
CAS#:87-41-2 CAS#:100-52-7
CAS#:100-52-7 CAS#:1483-71-2
CAS#:1483-71-2 CAS#:14303-28-7
CAS#:14303-28-7 CAS#:4505-48-0
CAS#:4505-48-0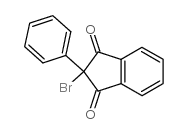 CAS#:1801-20-3
CAS#:1801-20-3 CAS#:1801-21-4
CAS#:1801-21-4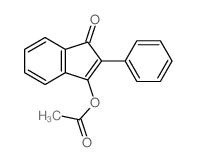 CAS#:1953-70-4
CAS#:1953-70-4 CAS#:520-03-6
CAS#:520-03-6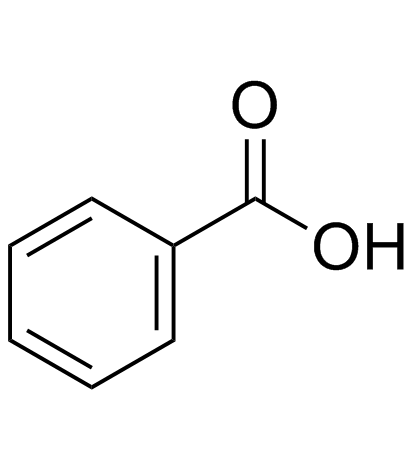 CAS#:65-85-0
CAS#:65-85-0 CAS#:84-66-2
CAS#:84-66-2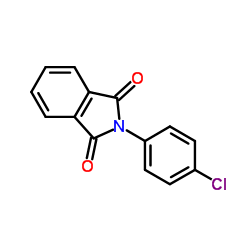 CAS#:7386-21-2
CAS#:7386-21-2
