氢化可的松-17-戊酸酯
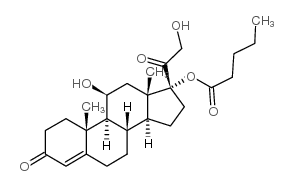
氢化可的松-17-戊酸酯结构式

|
常用名 | 氢化可的松-17-戊酸酯 | 英文名 | Hydrocortisone Valerate |
|---|---|---|---|---|
| CAS号 | 57524-89-7 | 分子量 | 446.57600 | |
| 密度 | 1.21g/cm3 | 沸点 | 595.1ºC at 760mmHg | |
| 分子式 | C26H38O6 | 熔点 | 217-220 °C | |
| MSDS | 中文版 美版 | 闪点 | 195ºC | |
| 符号 |

GHS08 |
信号词 | Warning |
|
Topical corticosteroid compounding: effects on physicochemical stability and skin penetration rate.
J. Am. Acad. Dermatol. 21(5 Pt 1) , 979-84, (1989) Compounding compatibility studies of four corticosteroid cream products and four commonly added chemicals are presented. Physical alteration, chemical stability, micropreservative challenge status, and in vitro skin penetration were evaluated at ambient condi... |
|
|
Addition of a topically applied corticosteroid to a modified Goeckerman regimen for treatment of psoriasis: effect on duration of remission.
J. Am. Acad. Dermatol. 13(5 Pt 1) , 784-91, (1985) A double-blind parallel group study was undertaken to assess the effect of adding a topically applied corticosteroid cream to a modified Goeckerman regimen to treat patients with psoriasis. Nineteen patients with psoriasis were treated with either this regime... |
|
|
Temporal infiltration of leukocyte subsets into mouse skin inflamed with phorbol ester.
Agents Actions 37(3-4) , 260-7, (1992) We have previously shown that multiple topical applications, over 11 days, of the phorbol ester 12-O-tetradecanoylphorbol-13-acetate (TPA) induces a persistent inflammatory reaction characterized by edema, cell infiltration and epidermal hyperplasia. In order... |
|
|
Contact hypersensitivity to tixocortol pivalate.
J. Am. Acad. Dermatol. 38(5 Pt 1) , 691-5, (1998) Tixocortol pivalate is an established marker to topical corticosteroid allergy. The prevalence of tixocortol pivalate hypersensitivity is well established in Europe, where exposure to this corticosteroid as a therapeutic agent varies. In the United States, ti... |
|
|
A dermatitis-eosinophilia syndrome. Treatment with methylprednisolone pulse therapy.
Arch. Dermatol. 120(12) , 1595-7, (1984) A 47-year-old man had a generalized, eczematous erythroderma and eosinophilia one week after a wasp sting. These changes persisted for four months despite intensive topical therapy and oral corticosteroids. He was then given corticosteroid pulse therapy with ... |
|
|
Contact allergic reaction to valerate esters of betamethasone and hydrocortisone.
Contact Dermatitis 12(1) , 58, (1985)
|
|
|
Clinical evaluation of hydrocortisone valerate 0.2% ointment.
Clin. Ther. 6(3) , 282-93, (1984) Evaluations of the comparative efficacy and safety of hydrocortisone valerate 0.2% ointment were made in six double-blind, multicenter trials involving a total of 485 patients, 209 with atopic dermatitis and 276 with plaque psoriasis. The vasoconstrictor acti... |
|
|
A comparison of once-daily application of mometasone furoate 0.1% cream compared with twice-daily hydrocortisone valerate 0.2% cream in pediatric atopic dermatitis patients who failed to respond to hydrocortisone: mometasone furoate study group.
Int. J. Dermatol. 38(8) , 604-6, (1999)
|
|
|
Clinical trials of topical corticosteroids in psoriasis: correlations with the vasoconstrictor assay.
Int. J. Dermatol. 31 Suppl 1 , 38-40, (1992) It would be desirable to develop an alternative system to clinical studies to evaluate the potency of generic topical corticosteroids and of new formulations of existing innovator corticosteroids. The vasoconstrictor assay is a reliable method for testing pot... |
|
|
Comparative efficacy of hydrocortisone valerate 0.2 percent ointment in the treatment of atopic dermatitis.
Cutis. 32(1) , 89-91, 94, (1983) Evaluations of the comparative efficacy and safety of a newly developed emollient ointment formulation of hydrocortisone valerate 0.2 percent were made in two double-blind, multicenter trials involving 145 patients with atopic dermatitis. Data are presented w... |