Cytidine
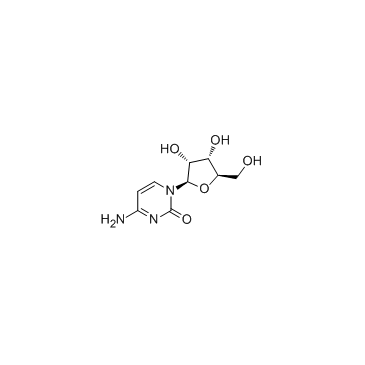
Cytidine structure
|
Common Name | Cytidine | ||
|---|---|---|---|---|
| CAS Number | 65-46-3 | Molecular Weight | 243.217 | |
| Density | 1.9±0.1 g/cm3 | Boiling Point | 529.7±60.0 °C at 760 mmHg | |
| Molecular Formula | C9H13N3O5 | Melting Point | 210-220 °C (dec.)(lit.) | |
| MSDS | Chinese USA | Flash Point | 274.1±32.9 °C | |
Use of CytidineCytidine is a nucleoside molecule that is formed when cytosine is attached to a ribose ring, cytidine is a component of RNA.Target: Nucleoside antimetabolite/analogCytidine is a nucleoside molecule that is formed when cytosine is attached to a ribose ring (also known as a ribofuranose) via a β-N1-glycosidic bond. Cytidine is a component of RNA. If cytosine is attached to a deoxyribose ring, it is known as a deoxycytidine. Dietary sources of cytidine include foods with high RNA (ribonucleic acid) content, such as organ meats, Brewer's yeast, as well as pyrimidine-rich foods such as beer. During digestion, RNA-rich foods are broken-down into ribosyl pyrimidines (cytidine and uridine), which are absorbed intact. In humans, dietary cytidine is converted into uridine, which is probably the compound behind cytidine's metabolic effects.There are a variety of cytidine analogs with potentially useful pharmacology. For example, KP-1461 is an anti-HIV agent that works as a viral mutagen, and zebularine exists in E. coli and is being examined for chemotherapy. Low doses of azacitidine and its analog decitabine have shown results against cancer through epigenetic demethylation. |
| Name | Cytidine |
|---|---|
| Synonym | More Synonyms |
| Description | Cytidine is a nucleoside molecule that is formed when cytosine is attached to a ribose ring, cytidine is a component of RNA.Target: Nucleoside antimetabolite/analogCytidine is a nucleoside molecule that is formed when cytosine is attached to a ribose ring (also known as a ribofuranose) via a β-N1-glycosidic bond. Cytidine is a component of RNA. If cytosine is attached to a deoxyribose ring, it is known as a deoxycytidine. Dietary sources of cytidine include foods with high RNA (ribonucleic acid) content, such as organ meats, Brewer's yeast, as well as pyrimidine-rich foods such as beer. During digestion, RNA-rich foods are broken-down into ribosyl pyrimidines (cytidine and uridine), which are absorbed intact. In humans, dietary cytidine is converted into uridine, which is probably the compound behind cytidine's metabolic effects.There are a variety of cytidine analogs with potentially useful pharmacology. For example, KP-1461 is an anti-HIV agent that works as a viral mutagen, and zebularine exists in E. coli and is being examined for chemotherapy. Low doses of azacitidine and its analog decitabine have shown results against cancer through epigenetic demethylation. |
|---|---|
| Related Catalog | |
| Target |
Human Endogenous Metabolite |
| References |
[1]. Jonas DA, et al. Safety considerations of DNA in food. Ann Nutr Metab. 2001;45(6):235-54. |
| Density | 1.9±0.1 g/cm3 |
|---|---|
| Boiling Point | 529.7±60.0 °C at 760 mmHg |
| Melting Point | 210-220 °C (dec.)(lit.) |
| Molecular Formula | C9H13N3O5 |
| Molecular Weight | 243.217 |
| Flash Point | 274.1±32.9 °C |
| Exact Mass | 243.085526 |
| PSA | 130.83000 |
| LogP | -1.78 |
| Vapour Pressure | 0.0±3.2 mmHg at 25°C |
| Index of Refraction | 1.756 |
| InChIKey | UHDGCWIWMRVCDJ-XVFCMESISA-N |
| SMILES | Nc1ccn(C2OC(CO)C(O)C2O)c(=O)n1 |
| Water Solubility | SOLUBLE |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Precursor 7 | |
|---|---|
| DownStream 10 | |
| HS Code | 2934999090 |
|---|---|
| Summary | 2934999090. other heterocyclic compounds. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Epigenetic variation in the Egfr gene generates quantitative variation in a complex trait in ants.
Nat. Commun. 6 , 6513, (2015) Complex quantitative traits, like size and behaviour, are a pervasive feature of natural populations. Quantitative trait variation is the product of both genetic and environmental factors, yet little ... |
|
|
The contribution of CMP kinase to the efficiency of DNA repair.
Cell Cycle 14(3) , 354-63, (2015) Cellular supply of deoxynucleoside triphosphates (dNTPs) is crucial for DNA replication and repair. In this study, we investigated the role of CMP/UMP kinase (CMPK), an enzyme catalyzes CDP formation,... |
|
|
Brain metastatic cancer cells release microRNA-181c-containing extracellular vesicles capable of destructing blood-brain barrier.
Nat. Commun. 6 , 6716, (2015) Brain metastasis is an important cause of mortality in breast cancer patients. A key event during brain metastasis is the migration of cancer cells through blood-brain barrier (BBB). However, the mole... |
|
Name: Luminescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: mu-type opioid receptor isoform MOR-1 [Homo sapiens]
External Id: OPRM1-OPRD1_AG_LUMI_1536_1X%ACT PRUN
|
|
Name: Primary qHTS assay for inhibitors of alpha-synuclein gene (SNCA) expression
Source: NCGC
External Id: SNCA-p-activity-luciferase
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: MMV: Compounds (1uM) were screened in a 72hr growth assay monitored by flow cytometry...
Source: ChEMBL
Target: Plasmodium falciparum
External Id: CHEMBL2448810
|
|
Name: Quantitative High-Throughput drug screen in 47 multiple myeloma cell lines against th...
Source: NCGC
Target: N/A
External Id: s-my-keats_OPM1-m4-1
|
|
Name: Quantitative High-Throughput drug screen in 47 multiple myeloma cell lines against th...
Source: NCGC
Target: N/A
External Id: s-my-OC1MY5-m4-1
|
|
Name: Quantitative High-Throughput drug screen in 47 multiple myeloma cell lines against th...
Source: NCGC
Target: N/A
External Id: s-my-KMS_34-m4-1
|
|
Name: Quantitative High-Throughput drug screen in 47 multiple myeloma cell lines against th...
Source: NCGC
Target: N/A
External Id: s-my-keats_L363-m4-1
|
|
Name: Quantitative High-Throughput drug screen in 47 multiple myeloma cell lines against th...
Source: NCGC
Target: N/A
External Id: s-my-keats_OCIMY7-m4-1
|
|
Name: Quantitative High-Throughput drug screen in 47 multiple myeloma cell lines against th...
Source: NCGC
Target: N/A
External Id: s-my-MM1R-m4-1
|
| 1-(b-D-Ribofuranosyl)-2-oxo-4-amino-1,2-dihydro-1,3-diazine |
| EINECS 200-610-9 |
| 4-Amino-1-[(2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)tetrahydro-2-furanyl]-2(1H)-pyrimidinone |
| 1-β-D-Ribofuranosylcytosine |
| 4-Amino-1-β-δ-ribofuranosyl-2(1H)-pyrimidinone |
| 4-Amino-1-(β-D-ribofuranosyl)pyrimidin-2(1H)-on |
| MFCD02181091 |
| Cytidine |
| 4-Amino-1-b-D-ribofuranosyl-2(1H)-pyrimidinone |
| Cyd |
| Cytidin |
| 1-β-D-ribofuranosyl-Cytosine |
| Citidine |
| 1-(b-δ-Ribofuranosyl)-2-oxo-4-amino-1,2-dihydro-1,3-diazine |
| d-cytidine |
| 1-β-δ-Ribofuranosylcytosine |
| CYTIDINE extrapure |
| 1-b-D-Ribofuranosylcytosine |
| 4-Imino-1-(β-D-ribofuranosyl)-1,4-dihydro-2-pyrimidinol |
 CAS#:56787-28-1
CAS#:56787-28-1 CAS#:5040-18-6
CAS#:5040-18-6 CAS#:800379-05-9
CAS#:800379-05-9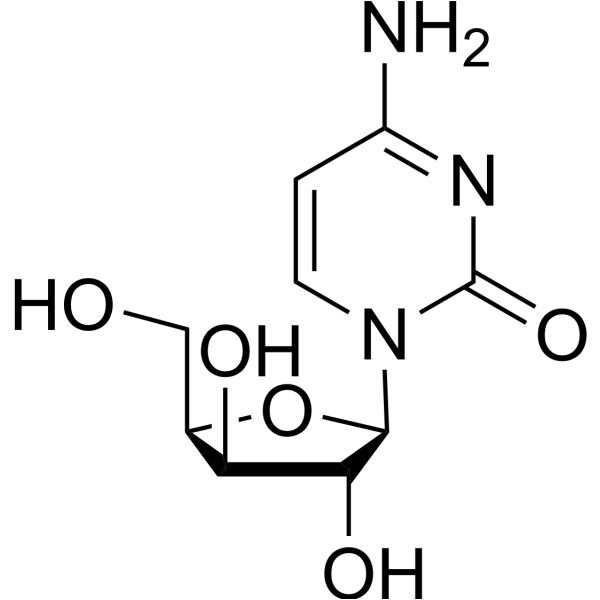 CAS#:3530-56-1
CAS#:3530-56-1 CAS#:23624-64-8
CAS#:23624-64-8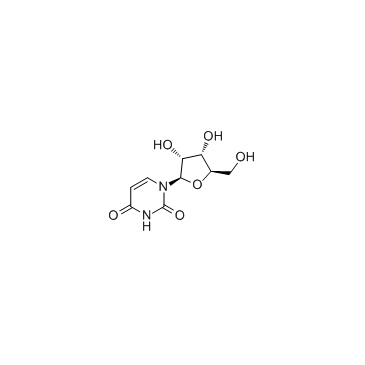 CAS#:58-96-8
CAS#:58-96-8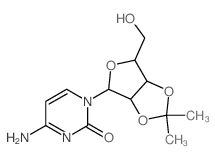 CAS#:362-42-5
CAS#:362-42-5 CAS#:3066-86-2
CAS#:3066-86-2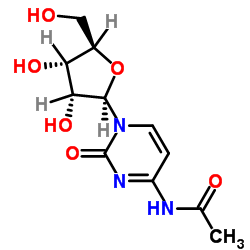 CAS#:3768-18-1
CAS#:3768-18-1 CAS#:31698-14-3
CAS#:31698-14-3 CAS#:50-69-1
CAS#:50-69-1 CAS#:71-30-7
CAS#:71-30-7 CAS#:36791-04-5
CAS#:36791-04-5 CAS#:4105-38-8
CAS#:4105-38-8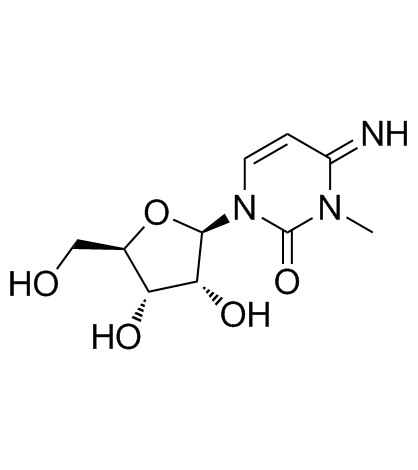 CAS#:2140-64-9
CAS#:2140-64-9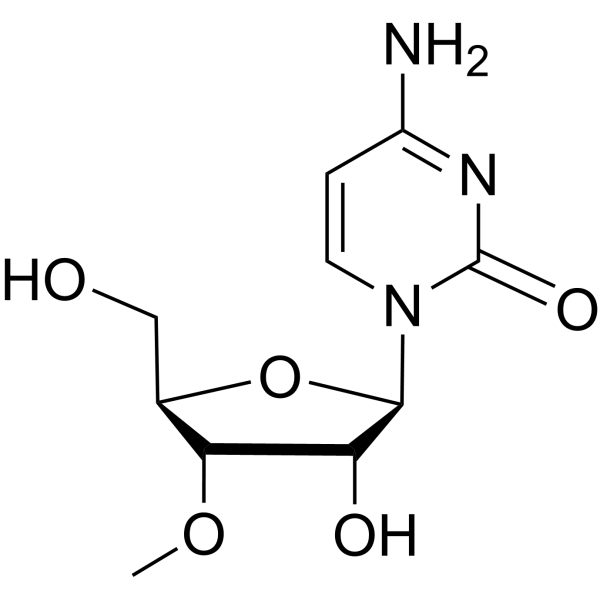 CAS#:20594-00-7
CAS#:20594-00-7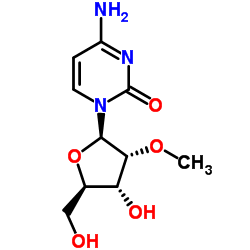 CAS#:2140-72-9
CAS#:2140-72-9
