Eudesmin
Modify Date: 2025-08-24 17:01:09
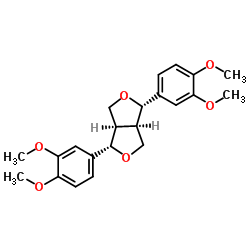
Eudesmin structure
|
Common Name | Eudesmin | ||
|---|---|---|---|---|
| CAS Number | 526-06-7 | Molecular Weight | 386.438 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 517.3±50.0 °C at 760 mmHg | |
| Molecular Formula | C22H26O6 | Melting Point | 108.5-109.5℃ | |
| MSDS | N/A | Flash Point | 209.8±30.0 °C | |
Use of EudesminEudesmin ((-)-Eudesmin) impairs adipogenic differentiation via inhibition of S6K1 signaling pathway. Eudesmin possesses diverse therapeutic effects, including anti-tumor, anti-inflammatory, and anti-bacterial activities[1]. |
| Name | 1H,3H-Furo[3,4-c]furan, 1,4-bis(3,4-dimethoxyphenyl)tetrahydro-, (1.α.,3a.α.,4.α.,6a.α.)-(-) |
|---|---|
| Synonym | More Synonyms |
| Description | Eudesmin ((-)-Eudesmin) impairs adipogenic differentiation via inhibition of S6K1 signaling pathway. Eudesmin possesses diverse therapeutic effects, including anti-tumor, anti-inflammatory, and anti-bacterial activities[1]. |
|---|---|
| Related Catalog | |
| Target |
S6K1[1] |
| In Vitro | Treatment of mesenchymal stem cells (MSCs) with Eudesmin (20, 40, and 80μM) disturbs adipogenesis via suppression of S6K1 signaling pathway. Eudesmin treatment inhibits activation and nuclear translocation of S6K1. S6K1-mediated phosphorylation of H2B at serine 36 (H2BS36p) is reduced upon Eudesmin treatment[1]. |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 517.3±50.0 °C at 760 mmHg |
| Melting Point | 108.5-109.5℃ |
| Molecular Formula | C22H26O6 |
| Molecular Weight | 386.438 |
| Flash Point | 209.8±30.0 °C |
| Exact Mass | 386.172943 |
| PSA | 55.38000 |
| LogP | 3.08 |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C |
| Index of Refraction | 1.547 |
| InChIKey | PEUUVVGQIVMSAW-UHFFFAOYSA-N |
| SMILES | COc1ccc(C2OCC3C(c4ccc(OC)c(OC)c4)OCC23)cc1OC |
| Storage condition | 2-8°C |
| Precursor 0 | |
|---|---|
| DownStream 3 | |
|
Name: Antifungal activity against Phomopsis obscurans assessed as growth inhibition at 50 u...
Source: ChEMBL
Target: Phomopsis obscurans
External Id: CHEMBL3052906
|
|
Name: Antifungal activity against Colletotrichum acutatum assessed as growth inhibition at ...
Source: ChEMBL
Target: Colletotrichum acutatum
External Id: CHEMBL3052908
|
|
Name: Antifungal activity against Colletotrichum gloeosporioides assessed as growth inhibit...
Source: ChEMBL
Target: Colletotrichum gloeosporioides
External Id: CHEMBL3052909
|
|
Name: Antifungal activity against Diaporthe ampelina assessed as growth inhibition at 50 to...
Source: ChEMBL
Target: Diaporthe ampelina
External Id: CHEMBL3052910
|
|
Name: Antifungal activity against Colletotrichum fragariae assessed as growth inhibition at...
Source: ChEMBL
Target: Colletotrichum fragariae
External Id: CHEMBL3052911
|
|
Name: Antifungal activity against Phomopsis obscurans assessed as growth inhibition at 50 t...
Source: ChEMBL
Target: Phomopsis obscurans
External Id: CHEMBL3052912
|
|
Name: Antifungal activity against Fusarium oxysporum assessed as growth inhibition at 50 to...
Source: ChEMBL
Target: Fusarium oxysporum
External Id: CHEMBL3052913
|
|
Name: Antialgal activity against Planktothrix agardhii assessed as inhibition measured for ...
Source: ChEMBL
Target: Planktothrix agardhii
External Id: CHEMBL3052942
|
|
Name: Small-molecule inhibitors of ST2 (IL1RL1)
Source: 20881
Target: interleukin-1 receptor-like 1 isoform [homo sapiens]
External Id: ST2_IL33_Inhibitors_Primary_Screening_77700
|
|
Name: Inhibition of LPS-induced nitric oxide production in mouse RAW264.7 cells after 24 hr...
Source: ChEMBL
Target: RAW264.7
External Id: CHEMBL3611158
|
Total 11, Current Page 1 of 2
1
2
| Pinoresinol dimethyl ether |
| Eudesamin,Eudesmin |
| (1S,3aR,4S,6aR)-1,4-Bis(3,4-dimethoxyphenyl)tetrahydro-1H,3H-furo[3,4-c]furan |
| (7S,7’S,8R,8’R)-eudesmin |
| (1S,3aR,4S,6aR)-1,4-bis(3,4-dimethoxyphenyl)-hexahydrofuro[3,4-c]furan |
| eudesmin |
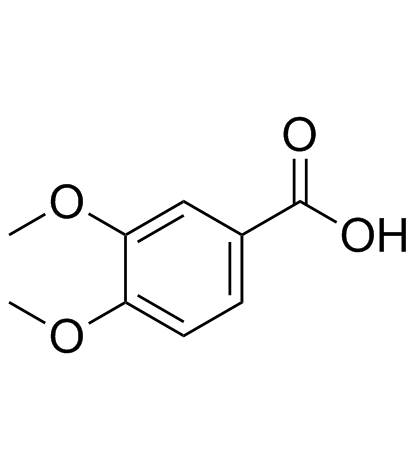 CAS#:93-07-2
CAS#:93-07-2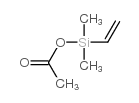 CAS#:3395-03-7
CAS#:3395-03-7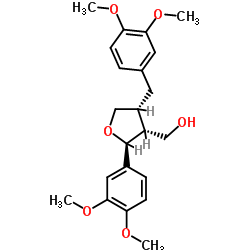 CAS#:67560-68-3
CAS#:67560-68-3
