5-Hydroxyindole-3-acetic acid
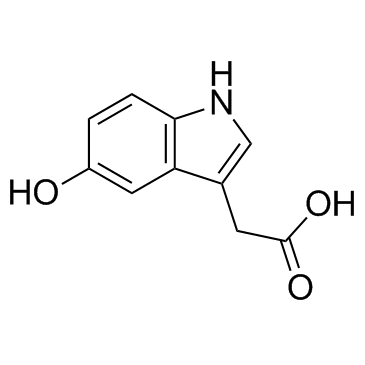
5-Hydroxyindole-3-acetic acid structure
|
Common Name | 5-Hydroxyindole-3-acetic acid | ||
|---|---|---|---|---|
| CAS Number | 54-16-0 | Molecular Weight | 191.183 | |
| Density | 1.496±0.06 g/cm3 | Boiling Point | 497.1±30.0 °C at 760 mmHg | |
| Molecular Formula | C10H9NO3 | Melting Point | 161-164ºC (dec.)(lit.) | |
| MSDS | USA | Flash Point | 254.4±24.6 °C | |
Use of 5-Hydroxyindole-3-acetic acid5-Hydroxyindole-3-acetic acid is the main metabolite of serotonin or metanephrines, which can be used as a biomarker of neuroendocrine tumors. |
| Name | (5-hydroxyindol-3-yl)acetic acid |
|---|---|
| Synonym | More Synonyms |
| Description | 5-Hydroxyindole-3-acetic acid is the main metabolite of serotonin or metanephrines, which can be used as a biomarker of neuroendocrine tumors. |
|---|---|
| Related Catalog | |
| Target |
Human Endogenous Metabolite |
| In Vitro | 5-Hydroxyindole-3-acetic acid (5HIAA) is a metabolite issued from serotonin metabolism, and the latter is synthetized from the essential amino acid tryptophan. Serotonin is mainly metabolised by monoamine oxidase in the liver and the lungs[1]. |
| References |
| Density | 1.496±0.06 g/cm3 |
|---|---|
| Boiling Point | 497.1±30.0 °C at 760 mmHg |
| Melting Point | 161-164ºC (dec.)(lit.) |
| Molecular Formula | C10H9NO3 |
| Molecular Weight | 191.183 |
| Flash Point | 254.4±24.6 °C |
| Exact Mass | 191.058243 |
| PSA | 73.32000 |
| LogP | 0.26 |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C |
| Index of Refraction | 1.740 |
| InChIKey | DUUGKQCEGZLZNO-UHFFFAOYSA-N |
| SMILES | O=C(O)Cc1c[nH]c2ccc(O)cc12 |
| Water Solubility | Sparingly soluble (11 g/L) (25 ºC) |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Safety Phrases | S22;S24/25 |
| RIDADR | NONH for all modes of transport |
| RTECS | NL3650000 |
| HS Code | 2933990090 |
|
~% 
5-Hydroxyindole... CAS#:54-16-0 |
| Literature: Journal of Organic Chemistry, , vol. 24, p. 179,183 Journal of the American Chemical Society, , vol. 76, p. 5579,5584 |
|
~% 
5-Hydroxyindole... CAS#:54-16-0 |
| Literature: Journal of Organic Chemistry, , vol. 24, p. 179,183 Journal of the American Chemical Society, , vol. 76, p. 5579,5584 |
|
~% 
5-Hydroxyindole... CAS#:54-16-0 |
| Literature: Journal of the American Chemical Society, , vol. 76, p. 5579,5584 |
|
~% 
5-Hydroxyindole... CAS#:54-16-0 |
| Literature: Journal of the American Chemical Society, , vol. 76, p. 5579,5584 |
|
~% 
5-Hydroxyindole... CAS#:54-16-0 |
| Literature: Farmaco, Edizione Scientifica, , vol. 11, p. 219 |
|
~% 
5-Hydroxyindole... CAS#:54-16-0 |
| Literature: Farmaco, Edizione Scientifica, , vol. 11, p. 219 |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Permeation of Dopamine Sulfate through the Blood-Brain Barrier.
PLoS ONE 10 , e0133904, (2015) Dopamine sulfate (DA-3- and DA-4-S) have been determined in the human brain, but it is unclear whether they are locally formed in the central nervous system (CNS), or transported into the CNS from per... |
|
|
Chemical genetics reveals a complex functional ground state of neural stem cells.
Nat. Chem. Biol. 3(5) , 268-273, (2007) The identification of self-renewing and multipotent neural stem cells (NSCs) in the mammalian brain holds promise for the treatment of neurological diseases and has yielded new insight into brain canc... |
|
|
The urinary ratio of 3-hydroxykynurenine/3-hydroxyanthranilic acid is an index to predicting the adverse effects of D-tryptophan in rats.
J. Nutr. Sci. Vitaminol. 60(4) , 261-8, (2014) The adverse effects of D-tryptophan and the possibility of it being a surrogate index for predicting adverse effects in rats were investigated. Male rats were fed one of several test diets (20% casein... |
| 5-Hydroxyindoleacetic acid |
| (5-Hydroxy-1H-indol-3-yl)acetic acid |
| 2-(5-Hydroxy-1H-indol-3-yl)acetic acid |
| (5-hydroxyindol-3-yl)acetic acid |
| 5-hydroxy-indole-3-acetic acid |
| EINECS 200-195-4 |
| MFCD00005639 |
| 5-Hydroxyindoleacetate |
| 5-Hydroxyindole-3-acetic Acid |
| 1H-Indole-3-acetic acid, 5-hydroxy- |
| 5-Hydroxyindolacetic Acid |

 CAS#:154-02-9
CAS#:154-02-9 CAS#:7313-69-1
CAS#:7313-69-1
