Trifluorothymidine
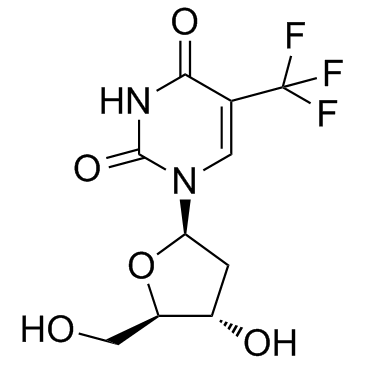
Trifluorothymidine structure
|
Common Name | Trifluorothymidine | ||
|---|---|---|---|---|
| CAS Number | 70-00-8 | Molecular Weight | 296.200 | |
| Density | 1.6±0.1 g/cm3 | Boiling Point | N/A | |
| Molecular Formula | C10H11F3N2O5 | Melting Point | 190-193 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | N/A | |
| Symbol |


GHS07, GHS08 |
Signal Word | Warning | |
Use of TrifluorothymidineTrifluridine is an irreversible thymidylate synthase inhibitor, and thereby suppresses DNA synthesis. Trifluridine is an antiviral drug for herpes simplex virus (HSV) infection. |
| Name | trifluridine |
|---|---|
| Synonym | More Synonyms |
| Description | Trifluridine is an irreversible thymidylate synthase inhibitor, and thereby suppresses DNA synthesis. Trifluridine is an antiviral drug for herpes simplex virus (HSV) infection. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.6±0.1 g/cm3 |
|---|---|
| Melting Point | 190-193 °C(lit.) |
| Molecular Formula | C10H11F3N2O5 |
| Molecular Weight | 296.200 |
| Exact Mass | 296.062012 |
| PSA | 104.55000 |
| LogP | 0.07 |
| Index of Refraction | 1.534 |
| InChIKey | VSQQQLOSPVPRAZ-RRKCRQDMSA-N |
| SMILES | O=c1[nH]c(=O)n(C2CC(O)C(CO)O2)cc1C(F)(F)F |
| Storage condition | −20°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |


GHS07, GHS08 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302 + H312 + H332-H351 |
| Precautionary Statements | P261-P280-P301 + P312 + P330 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi:Irritant |
| Risk Phrases | R20/21/22;R40 |
| Safety Phrases | S22-S36 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | XP2087500 |
| Precursor 8 | |
|---|---|
| DownStream 2 | |
|
Mechanistic evaluation of Ginkgo biloba leaf extract-induced genotoxicity in L5178Y cells.
Toxicol. Sci. 139(2) , 338-49, (2014) Ginkgo biloba has been used for many thousand years as a traditional herbal remedy and its extract has been consumed for many decades as a dietary supplement. Ginkgo biloba leaf extract is a complex m... |
|
|
Quantitative high-throughput identification of drugs as modulators of human constitutive androstane receptor.
Sci. Rep. 5 , 10405, (2015) The constitutive androstane receptor (CAR, NR1I3) plays a key role in governing the transcription of numerous hepatic genes that involve xenobiotic metabolism/clearance, energy homeostasis, and cell p... |
|
|
Differential activation of cell death and autophagy results in an increased cytotoxic potential for trifluorothymidine compared to 5-fluorouracil in colon cancer cells.
Int. J. Cancer 126 , 2457-2468, (2010) Trifluorothymidine (TFT) is part of the oral drug formulation TAS-102. Both 5-fluorouracil (5-FU) and TFT can inhibit thymidylate synthase and be incorporated into DNA. TFT shows only moderate cross-r... |
|
Name: Primary qHTS assay for inhibitors of alpha-synuclein gene (SNCA) expression
Source: NCGC
External Id: SNCA-p-activity-luciferase
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify ago...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_AG_FLUO8_1536_1X%ACT PRUN
|
|
Name: Cytochrome P450 Family 1 Subfamily A Member 2 (CYP1A2) small molecule antagonists: lu...
Source: 824
External Id: CYP273
|
|
Name: Evaluation of antiviral activity against Vaccinia virus
Source: ChEMBL
Target: Vaccinia virus
External Id: CHEMBL816930
|
|
Name: Fluorescence-based cell-based primary high throughput screening assay to identify pos...
Source: The Scripps Research Institute Molecular Screening Center
Target: muscarinic acetylcholine receptor M1 [Homo sapiens]
External Id: CHRM1_PAM_FLUO8_1536_1X%ACT PRUN
|
|
Name: Antiviral activity against HSV1 CL-101 infected in African green monkey Vero cells as...
Source: ChEMBL
Target: NON-PROTEIN TARGET
External Id: CHEMBL3282724
|
|
Name: Cytotoxicity against African green monkey Vero cells at 1 uM after 3 days by haemocyt...
Source: ChEMBL
Target: Vero
External Id: CHEMBL3282727
|
|
Name: Cytotoxicity against mouse Sarcoma 180 cells after 1 to 3 days by coulter counter ana...
Source: ChEMBL
Target: Sarcoma-180
External Id: CHEMBL3282729
|
|
Name: qHTS Assay for Small Molecule Inhibitors of the Human hERG Channel Activity
Source: NCGC
External Id: HERG01
|
|
Name: qHTS for Inhibitors of TGF-b: Cytotox Counterscreen
Source: NCGC
Target: N/A
External Id: SMAD3201
|
| 1-[(2R,4S,5R)-4-hydroxy-5-(hydroxyméthyl)tétrahydrofuran-2-yl]-5-(trifluorométhyl)pyrimidine-2,4(1H,3H)-dione |
| Viroptic |
| α,α,α-Trifluorothymidine |
| 2'-Deoxy-5-(trifluoromethyl)uridine |
| UNII-RMW9V5RW38 |
| Trifluorothymidine |
| 1-(2-Deoxy-β-D-glycero-pentofuranosyl)-5-(trifluoromethyl)pyrimidine-2,4(1H,3H)-dione |
| Trifluridine |
| 1-[(2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl]-5-(trifluoromethyl)pyrimidine-2,4(1H,3H)-dione |
| EINECS 200-722-8 |
| 1-[(2R,4S,5R)-4-Hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl]-5-(trifluormethyl)pyrimidin-2,4(1H,3H)-dion |
| 2'-Deoxy-5-trifluoromethyluridine |
| Virophta |
| a,a,a-Trifluorothymidine |
| MFCD00006534 |
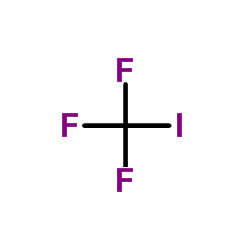 CAS#:2314-97-8
CAS#:2314-97-8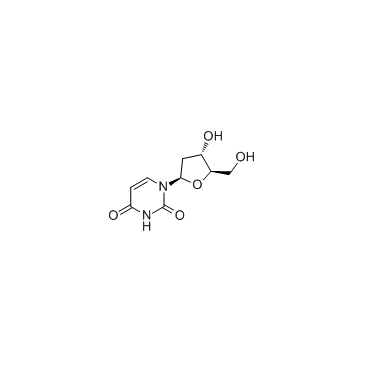 CAS#:951-78-0
CAS#:951-78-0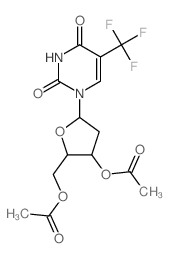 CAS#:65499-42-5
CAS#:65499-42-5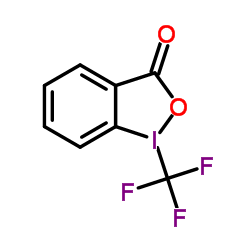 CAS#:887144-94-7
CAS#:887144-94-7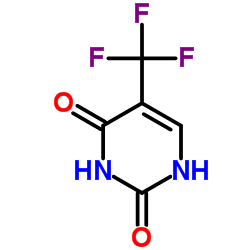 CAS#:54-20-6
CAS#:54-20-6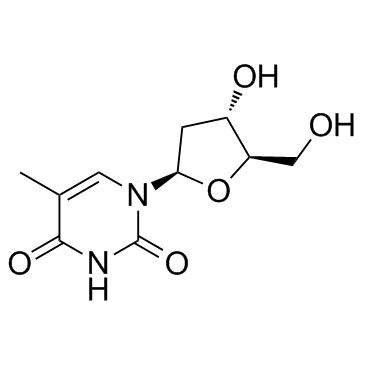 CAS#:50-89-5
CAS#:50-89-5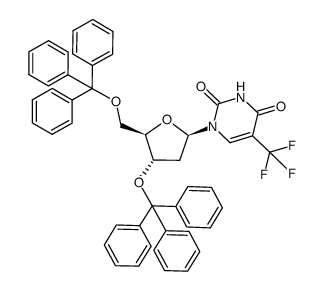 CAS#:76513-99-0
CAS#:76513-99-0 CAS#:13030-62-1
CAS#:13030-62-1 CAS#:14599-46-3
CAS#:14599-46-3![[5-[2,4-dioxo-5-(trifluoromethyl)pyrimidin-1-yl]-3-(4-methylbenzoyl)oxy-oxolan-2-yl]methyl 4-methylbenzoate structure](https://image.chemsrc.com/caspic/066/7057-46-7.png) CAS#:7057-46-7
CAS#:7057-46-7
