allantoin
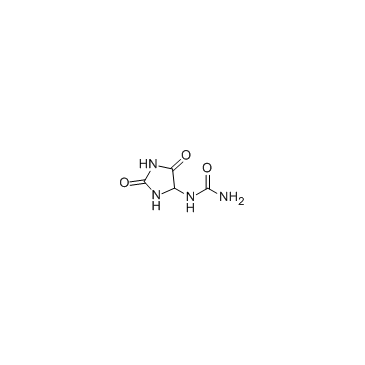
allantoin structure
|
Common Name | allantoin | ||
|---|---|---|---|---|
| CAS Number | 97-59-6 | Molecular Weight | 158.115 | |
| Density | 1.7±0.1 g/cm3 | Boiling Point | 478ºC | |
| Molecular Formula | C4H6N4O3 | Melting Point | 230 °C (dec.)(lit.) | |
| MSDS | Chinese USA | Flash Point | 230-234°C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of allantoinAllantoin is a skin conditioning agent that promotes healthy skin, stimulates new and healthy tissue growth. |
| Name | allantoin |
|---|---|
| Synonym | More Synonyms |
| Description | Allantoin is a skin conditioning agent that promotes healthy skin, stimulates new and healthy tissue growth. |
|---|---|
| Related Catalog | |
| Target |
Human Endogenous Metabolite |
| In Vitro | Allantoin is a well-known cosmetic ingredient reported to have anti-oxidative and anti-inflammatory activities[1]. Allantoin attenuates apoptosis and cytotoxicity and increased the viability of STZ-induced β-cells in a dose-dependent manner. Allantoin decreases the level of caspase-3 and increases the level of phosphorylated B-cell lymphoma 2 (Bcl-2) expression. Allantoin has been demonstrated to activate imidazoline 3 (I3) receptors[2]. |
| In Vivo | The subchronic administration of allantoin (1, 3 or 10 mg/kg, for 7 days) significantly increases the latency time measured during the passive avoidance task in scopolamine-induced cholinergic blockade and normal naive mice. Allantoin treatment (3 or 10 mg/kg, for 7 days) also increases the expression levels of phosphorylated phosphatidylinositide 3-kinase (PI3K), phosphorylated protein kinase B (Akt) and phosphorylated glycogen synthase kinase-3β (GSK-3β). Allantoin significantly increases the neuronal cell proliferation of immature neurons in the hippocampal dentate gyrus region[1]. Daily injection of allantoin for 8 days in STZ-treated rats significantly lowers plasma glucose and increases plasma insulin levels [2]. Allantoin decreases blood pressures in SHRs at 30 minutes, as the most effective time. Also, this antihypertensive action is shown in a dose-dependent manner from SHRs treated with allantoin. Moreover, in anesthetized rats, allantoin inhibits cardiac contractility and heart rate. Also, the peripheral blood flow is markedly increased by allantoin[3]. |
| Cell Assay | Pancreatic β-cells are treated with 1, 10, 100 μM of allantoin before 30 min prior to the addition of 5 mM STZ and incubated for 6 h. Cell viability is measured using the ApoTox-Glo triplex assay[2]. |
| Animal Admin | Rats: Animals are randomly assigned into four groups: (I) the control group treated with the vehicle, saline; (II) the allantoin group treated by intravenous injection of allantoin at 0.5 mg/kg; (III) the allantoin+efaroxan group treated with allantoin at the most effective dose (0.5 mg/kg, i.v.) and efaroxan at effective dose (1.5 mg/kg, i.v.) 30 minutes before injection of allantoin; and (IV) the allantoin treated SHRs group treated by intravenous injection of allantoin at various dose for desired time. After treatment of allantoin, the rats are placed into a holder for the determination of the mean blood pressure[3]. Mice: For memory ameliorating study, mice are administered vehicle solution, allantoin (1, 3 or 10 mg/kg, p.o.) or donepezil (5 mg/kg, p.o.) at the same time (10:00-12:00 a.m) and same place for 7 days. For memory enhancing study, mice are administered vehicle solution, allantoin (1, 3 or 10 mg/kg, p.o.) or piracetam (200 mg/kg, i.p.). The final administration of allantoin, donepezil or piracetam is performed 1 h before an acquisition trial in the passive avoidance task[1]. |
| References |
[3]. Chen MF, et al. Antihypertensive action of allantoin in animals. Biomed Res Int. 2014;2014:690135. |
| Density | 1.7±0.1 g/cm3 |
|---|---|
| Boiling Point | 478ºC |
| Melting Point | 230 °C (dec.)(lit.) |
| Molecular Formula | C4H6N4O3 |
| Molecular Weight | 158.115 |
| Flash Point | 230-234°C |
| Exact Mass | 158.043991 |
| PSA | 113.32000 |
| LogP | -2.89 |
| Index of Refraction | 1.616 |
| InChIKey | POJWUDADGALRAB-UHFFFAOYSA-N |
| SMILES | NC(=O)NC1NC(=O)NC1=O |
| Storage condition | Refrigerator |
CHEMICAL IDENTIFICATION
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302 |
| Precautionary Statements | P301 + P312 + P330 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn,Xi |
| Risk Phrases | R22 |
| Safety Phrases | S22-S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 1 |
| RTECS | YT1600000 |
| HS Code | 2933790090 |
| Precursor 10 | |
|---|---|
| DownStream 9 | |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Cleavage efficient 2A peptides for high level monoclonal antibody expression in CHO cells.
MAbs 7(2) , 403-12, (2015) Linking the heavy chain (HC) and light chain (LC) genes required for monoclonal antibodies (mAb) production on a single cassette using 2A peptides allows control of LC and HC ratio and reduces non-exp... |
|
|
Cheminformatics analysis of assertions mined from literature that describe drug-induced liver injury in different species.
Chem. Res. Toxicol. 23 , 171-83, (2010) Drug-induced liver injury is one of the main causes of drug attrition. The ability to predict the liver effects of drug candidates from their chemical structures is critical to help guide experimental... |
|
|
¹H-NMR and MS based metabolomics study of the intervention effect of curcumin on hyperlipidemia mice induced by high-fat diet.
PLoS ONE 10(3) , e0120950, (2015) Curcumin, a principle bioactive component of Curcuma longa L, is well known for its anti-hyperlipidemia effect. However, no holistic metabolic information of curcumin on hyperlipidemia models has been... |
| Allantol |
| DL-Allantoin |
| uniderma |
| ALL |
| Allantoin |
| Septalan |
| N-(2,5-Dioxo-4-imidazolidinyl)urea |
| 2,5-Dioxo-4-imidazolidinyl-urea |
| Urea, (2,5-dioxo-4-imidazolidinyl)- (9CI) |
| 4-ureido-2,5-Imidazolidinedione |
| toin |
| 5-ureidohydantoin |
| ALANTOIN |
| (±)-Allantoin |
| Sebical |
| Alantan |
| 1-(2,5-Dioxoimidazolidin-4-yl)harnstoff |
| Allantoin (8CI) |
| 1-(2,5-dioxoimidazolidin-4-yl)urea |
| 2,5-dioxo-4-imidazolidinyl urea |
| 1-(2,5-Dioxo-4-imidazolidinyl)urea |
| MFCD00005260 |
| Psoralon |
| 5-Ureido-2,4-imidazolidindion |
| 5-ureido-Hydantoin |
| EINECS 202-592-8 |
 CAS#:69-93-2
CAS#:69-93-2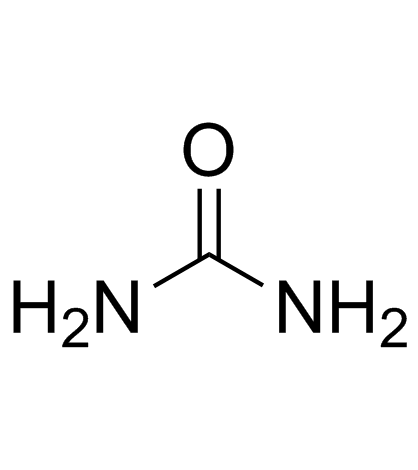 CAS#:57-13-6
CAS#:57-13-6 CAS#:298-12-4
CAS#:298-12-4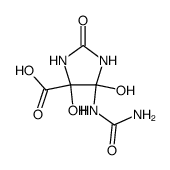 CAS#:874531-63-2
CAS#:874531-63-2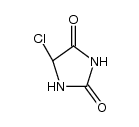 CAS#:32282-43-2
CAS#:32282-43-2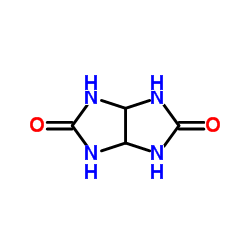 CAS#:496-46-8
CAS#:496-46-8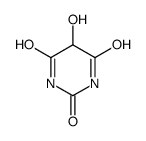 CAS#:444-15-5
CAS#:444-15-5 CAS#:99-16-1
CAS#:99-16-1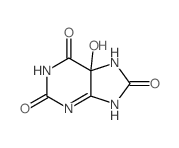 CAS#:6960-30-1
CAS#:6960-30-1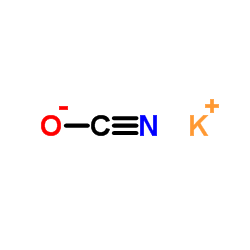 CAS#:590-28-3
CAS#:590-28-3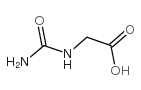 CAS#:462-60-2
CAS#:462-60-2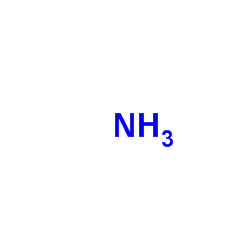 CAS#:7664-41-7
CAS#:7664-41-7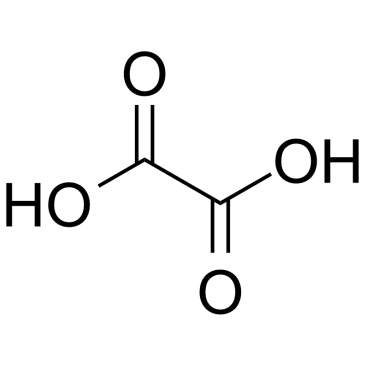 CAS#:144-62-7
CAS#:144-62-7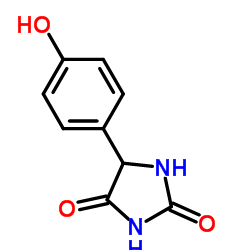 CAS#:2420-17-9
CAS#:2420-17-9
