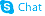Ne-Boc-L-lysine
Ne-Boc-L-lysine structure
|
Common Name | Ne-Boc-L-lysine | ||
|---|---|---|---|---|
| CAS Number | 2418-95-3 | Molecular Weight | 246.303 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 412.9±40.0 °C at 760 mmHg | |
| Molecular Formula | C11H22N2O4 | Melting Point | 250 °C (dec.)(lit.) | |
| MSDS | Chinese USA | Flash Point | 203.5±27.3 °C | |
Use of Ne-Boc-L-lysine(S)-2-Amino-6-((tert-butoxycarbonyl)amino)hexanoic acid is a lysine derivative[1]. |
| Name | Ne-Boc-L-lysine |
|---|---|
| Synonym | More Synonyms |
| Description | (S)-2-Amino-6-((tert-butoxycarbonyl)amino)hexanoic acid is a lysine derivative[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Amino acids and amino acid derivatives have been commercially used as ergogenic supplements. They influence the secretion of anabolic hormones, supply of fuel during exercise, mental performance during stress related tasks and prevent exercise induced muscle damage. They are recognized to be beneficial as ergogenic dietary substances[1]. |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 412.9±40.0 °C at 760 mmHg |
| Melting Point | 250 °C (dec.)(lit.) |
| Molecular Formula | C11H22N2O4 |
| Molecular Weight | 246.303 |
| Flash Point | 203.5±27.3 °C |
| Exact Mass | 246.157959 |
| PSA | 101.65000 |
| LogP | 0.74 |
| Vapour Pressure | 0.0±2.1 mmHg at 25°C |
| Index of Refraction | 1.485 |
| Storage condition | 2~8°C |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 2924199090 |
| Precursor 10 | |
|---|---|
| DownStream 9 | |
| HS Code | 2924199090 |
|---|---|
| Summary | 2924199090. other acyclic amides (including acyclic carbamates) and their derivatives; salts thereof. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Adding l-lysine derivatives to the genetic code of mammalian cells with engineered pyrrolysyl-tRNA synthetases.
Biochem. Biophys. Res. Commun. 371(4) , 818-22, (2008) We report a method for site-specifically incorporating l-lysine derivatives into proteins in mammalian cells, based on the expression of the pyrrolysyl-tRNA synthetase (PylRS)-tRNA(Pyl) pair from Meth... |
|
|
Evidence that malondialdehyde-derived aminoenimine is not a fluorescent age pigment.
Chem. Res. Toxicol. 14(5) , 473-5, (2001) It has been suggested that protein modifications by malondialdehyde (MDA), a major product of lipid peroxidation, contribute to the fluorescence formation of lipofuscin. Although early studies propose... |
|
|
Nanoassembly of surfactants with interfacial drug-interactive motifs as tailor-designed drug carriers.
Mol. Pharm. 10(1) , 187-98, (2013) PEGylated lipopeptide surfactants carrying drug-interactive motifs specific for a peptide-nitroxide antioxidant, JP4-039, were designed and constructed to facilitate the solubilization of this drug ca... |
| Z-Lys(Boc)-OH |
| Ne-Boc-L-Lysine |
| NEPSILON-BOC-L-LYSINE |
| N-[(Benzyloxy)carbonyl]-N-{[(2-methyl-2-propanyl)oxy]carbonyl}lysine |
| N2-BENZYLOXYCARBONYL-N6-BOC-L-LYSINE |
| e-BOC-L-lysine. |
| MFCD00037221 |
| N-{[(2-Methyl-2-propanyl)oxy]carbonyl}-L-lysine |
| H-L-LYS(BOC)-OH |
| L-Lysine, N-[(1,1-dimethylethoxy)carbonyl]- |
| L-LYSINE(BOC) |
| LYS(BOC) |
| Fmoc-Lys(Boc)-OH |
| Lysine derivative 1 |
| N-(Tert-Butoxycarbonyl)-L-Lysine |
| Nε-Boc-L-lysine |
| N2-Benzyloxycarbonyl-N6-tert-butyloxycarbonyl-L-lysine |
| L-LYS(BOC) |
| Nε-Boc-Nα-Cbz-L-lysine |
| N6-Boc-L-lysine |
| Nepsilon-Boc-Nalpha-Cbz-L-Lysine |
| Nε-(tert-Butoxycarbonyl)-L-lysine |
| (S)-2-Amino-6-((tert-butoxycarbonyl)amino)hexanoic acid |
| LYSINE(BOC)-OH |
| (S)-2-(((Benzyloxy)carbonyl)amino)-6-((tert-butoxycarbonyl)amino)hexanoic acid |
| N2-benzyloxycarbonyl-N6-tert-butoxycarbonyl-L-lysine |
| H-Lys(Boc)-OH |
| L-Lysine, N6-((1,1-dimethylethoxy)carbonyl)- |
| AmbotzZAA1184 |
| Lysine, N-[(1,1-dimethylethoxy)carbonyl]-N-[(phenylmethoxy)carbonyl]- |
| N-Cbz-N'-Boc-L-lysine |
| Nε-(tert-Butoxycarbonyl)-Nα-carbobenzoxy-L-lysine |
 CAS#:2389-60-8
CAS#:2389-60-8 CAS#:24424-99-5
CAS#:24424-99-5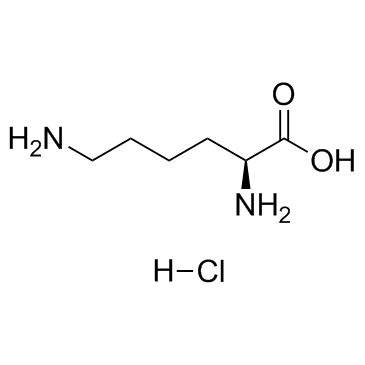 CAS#:657-27-2
CAS#:657-27-2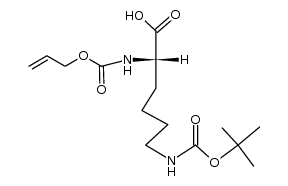 CAS#:104669-72-9
CAS#:104669-72-9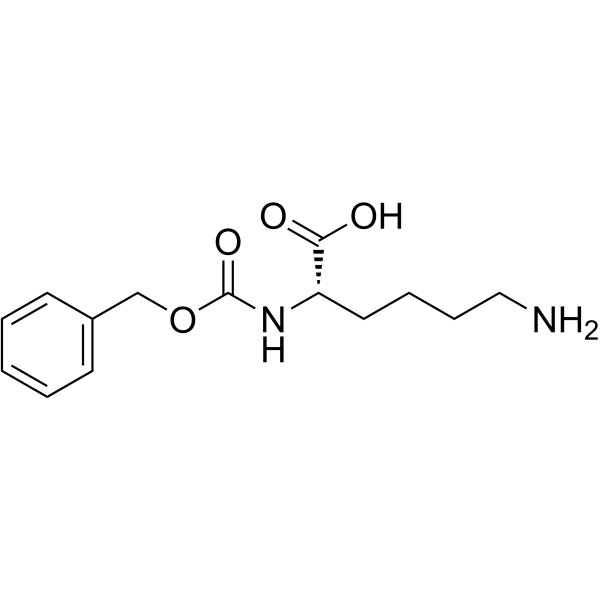 CAS#:2212-75-1
CAS#:2212-75-1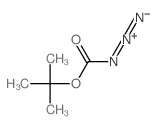 CAS#:1070-19-5
CAS#:1070-19-5 CAS#:56-87-1
CAS#:56-87-1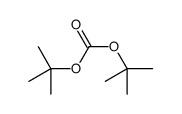 CAS#:34619-03-9
CAS#:34619-03-9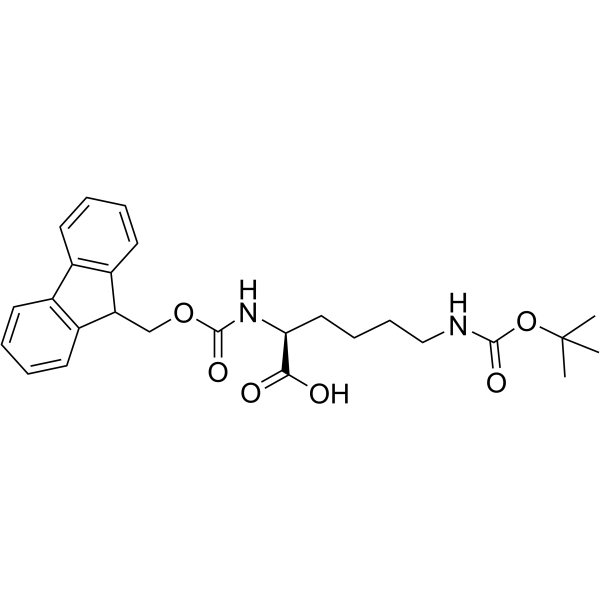 CAS#:71989-26-9
CAS#:71989-26-9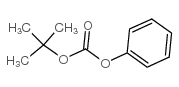 CAS#:6627-89-0
CAS#:6627-89-0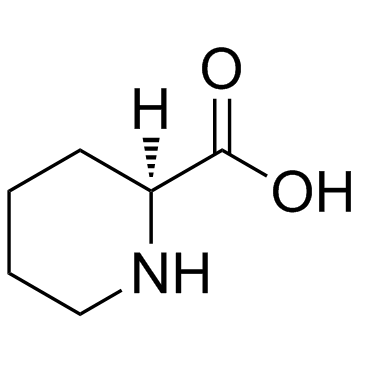 CAS#:3105-95-1
CAS#:3105-95-1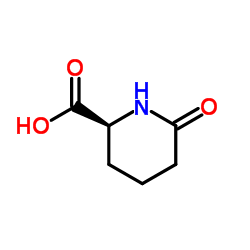 CAS#:34622-39-4
CAS#:34622-39-4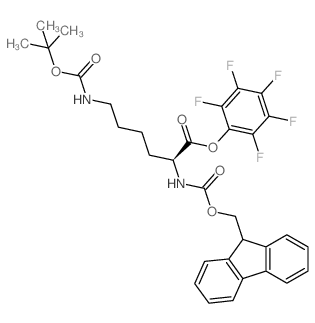 CAS#:86060-98-2
CAS#:86060-98-2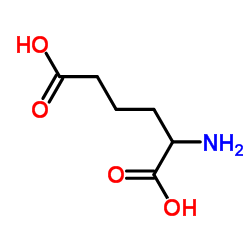 CAS#:1118-90-7
CAS#:1118-90-7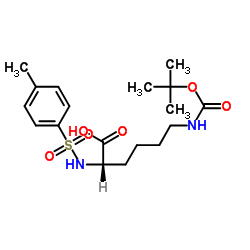 CAS#:16948-09-7
CAS#:16948-09-7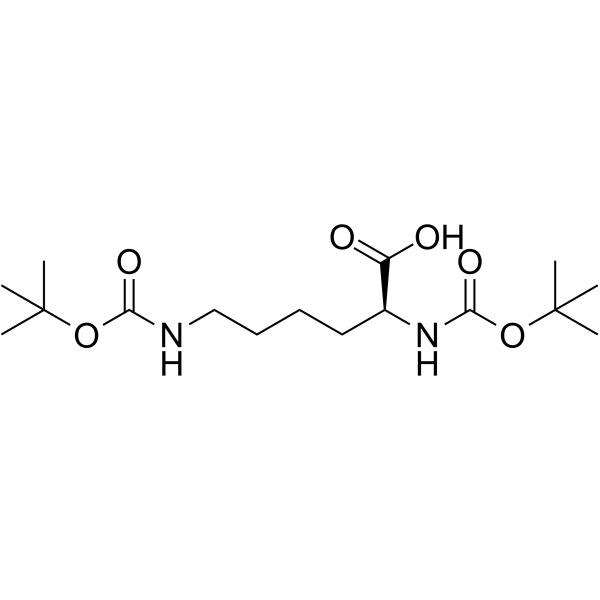 CAS#:2483-46-7
CAS#:2483-46-7