Tetrazine-Ph-NHCO-C3-NHS ester
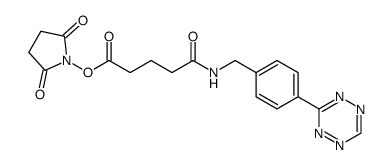
Tetrazine-Ph-NHCO-C3-NHS ester structure
|
Common Name | Tetrazine-Ph-NHCO-C3-NHS ester | ||
|---|---|---|---|---|
| CAS Number | 1244040-64-9 | Molecular Weight | 398.37300 | |
| Density | N/A | Boiling Point | N/A | |
| Molecular Formula | C18H18N6O5 | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | N/A | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of Tetrazine-Ph-NHCO-C3-NHS esterTetrazine-Ph-NHCO-C3-NHS ester is a PEG-based PROTAC linker that can be used in the synthesis of PROTACs[1]. |
| Name | 2,5-dioxopyrrolidin-1-yl 5-((4-(1,2,4,5-tetrazin-3-yl)benzyl)amino)-5-oxopentanoate |
|---|
| Description | Tetrazine-Ph-NHCO-C3-NHS ester is a PEG-based PROTAC linker that can be used in the synthesis of PROTACs[1]. |
|---|---|
| Related Catalog | |
| Target |
PEGs |
| In Vitro | PROTACs contain two different ligands connected by a linker; one is a ligand for an E3 ubiquitin ligase and the other is for the target protein. PROTACs exploit the intracellular ubiquitin-proteasome system to selectively degrade target proteins[1]. |
| References |
| Molecular Formula | C18H18N6O5 |
|---|---|
| Molecular Weight | 398.37300 |
| Exact Mass | 398.13400 |
| PSA | 144.34000 |
| LogP | 0.65610 |
| InChIKey | QDUUJWMNSUXICN-UHFFFAOYSA-N |
| SMILES | O=C(CCCC(=O)ON1C(=O)CCC1=O)NCc1ccc(-c2nncnn2)cc1 |
|
Synthesis and evaluation of a series of 1,2,4,5-tetrazines for bioorthogonal conjugation.
Bioconjug. Chem. 22 , 2263-2270, (2011) 1,2,4,5-Tetrazines have been established as effective dienes for inverse electron demand [4 + 2] Diels-Alder cycloaddition reactions with strained alkenes for over 50 years. Recently, this reaction pa... |
|
|
Tetrazine-based cycloadditions: application to pretargeted live cell imaging.
Bioconjug. Chem. 19 , 2297-2299, (2008) Bioorthogonal tetrazine cycloadditions have been applied to live cell labeling. Tetrazines react irreversibly with the strained dienophile norbornene forming dihydropyrazine products and dinitrogen. T... |
|
|
Tetrazine ligation: fast bioconjugation based on inverse-electron-demand Diels-Alder reactivity.
J. Am. Chem. Soc. 130 , 13518-13519, (2008) Described is a bioorthogonal reaction that proceeds with unusually fast reaction rates without need for catalysis: the cycloaddition of s-tetrazine and trans-cyclooctene derivatives. The reactions tol... |